5437-48-9
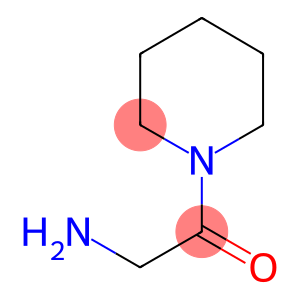
2-oxo-2-piperidin-1-ylethanamine hydrochloride
CAS: 5437-48-9
Molecular Formula: C7H15ClN2O
5437-48-9 - Names and Identifiers
5437-48-9 - Physico-chemical Properties
| Molecular Formula | C7H15ClN2O |
| Molar Mass | 178.66 |
| Boling Point | 311.2°C at 760 mmHg |
| Flash Point | 142°C |
| Vapor Presure | 0.000422mmHg at 25°C |
| Storage Condition | Sealed in dry,Room Temperature |
5437-48-9 - Risk and Safety
| Hazard Class | IRRITANT |
5437-48-9 - Introduction
hydrochloride, also known as hydrochloride, is an organic compound. The following is a description of its nature, use, preparation and safety information:
1. nature:
-Chemical formula: C8H16ClNO
-Molecular weight: 167.674g/mol
-Appearance: White crystalline solid
-Melting Point: 128-131°C
-Soluble: Soluble in water and ethanol
2. use:
-As a drug intermediate: hydrochloride can be used to synthesize a variety of drugs, such as anti-anxiety drugs and analgesic drugs.
-Scientific research use: It is also commonly used in organic synthesis reactions as a catalyst or reducing agent.
3. Preparation method:
-hydrochloride can be obtained by reacting 2-amino-1-piperidin-1-one with acetaldehyde under acid-catalyzed conditions. Then by the addition of hydrochloric acid, hydrochloride can be obtained.
4. Safety Information:
-hydrochloride is generally a low-toxic compound, but it is still necessary to pay attention to reasonable use and correct operation and storage.
-Follow laboratory safety regulations when using, avoid contact with skin and eyes, and wear personal protective equipment such as lab gloves and goggles during use.
-Avoid inhaling the dust or gas of the compound and ensure that the operation is carried out in a well-ventilated environment.
-If the compound is accidentally taken or exposed, seek medical help immediately and provide information about the compound to your doctor.
1. nature:
-Chemical formula: C8H16ClNO
-Molecular weight: 167.674g/mol
-Appearance: White crystalline solid
-Melting Point: 128-131°C
-Soluble: Soluble in water and ethanol
2. use:
-As a drug intermediate: hydrochloride can be used to synthesize a variety of drugs, such as anti-anxiety drugs and analgesic drugs.
-Scientific research use: It is also commonly used in organic synthesis reactions as a catalyst or reducing agent.
3. Preparation method:
-hydrochloride can be obtained by reacting 2-amino-1-piperidin-1-one with acetaldehyde under acid-catalyzed conditions. Then by the addition of hydrochloric acid, hydrochloride can be obtained.
4. Safety Information:
-hydrochloride is generally a low-toxic compound, but it is still necessary to pay attention to reasonable use and correct operation and storage.
-Follow laboratory safety regulations when using, avoid contact with skin and eyes, and wear personal protective equipment such as lab gloves and goggles during use.
-Avoid inhaling the dust or gas of the compound and ensure that the operation is carried out in a well-ventilated environment.
-If the compound is accidentally taken or exposed, seek medical help immediately and provide information about the compound to your doctor.
Last Update:2024-04-09 15:17:58
Supplier List
Spot supply
Product Name: 2-Amino-1-(1-piperidinyl)-ethanone HCl Visit Supplier Webpage Request for quotationCAS: 5437-48-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-AMINO-1-PIPERIDIN-1-YL-ETHANONE HCL Request for quotation
CAS: 5437-48-9
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 5437-48-9
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: 2-AMINO-1-PIPERIDIN-1-YL-ETHANONE HCL Visit Supplier Webpage Request for quotation
CAS: 5437-48-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 5437-48-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 2-Amino-1-(1-piperidinyl)-ethanone HCl Visit Supplier Webpage Request for quotationCAS: 5437-48-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-AMINO-1-PIPERIDIN-1-YL-ETHANONE HCL Request for quotation
CAS: 5437-48-9
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 5437-48-9
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: 2-AMINO-1-PIPERIDIN-1-YL-ETHANONE HCL Visit Supplier Webpage Request for quotation
CAS: 5437-48-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 5437-48-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


