54143-55-4
Flecanide
CAS: 54143-55-4
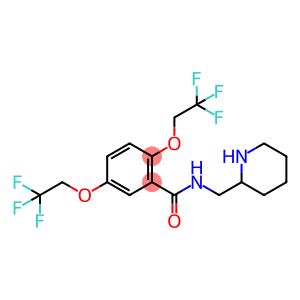
Molecular Formula: C17H20F6N2O3
54143-55-4 - Names and Identifiers
| Name | Flecanide |
| Synonyms | Flecaine Flecanide Flecainide Flecainide-d4 rac Flecainide FLECAINTDE BASE -2,5-bis(2,2,2-trifluoroethoxy) N-(2-Piperidinylmethyl)-2,5-bis(2,2,2-trifluoroethoxy)benzamide N-(Piperidin-2-ylMethyl)-2,5-bis(2,2,2-trifluoroethoxy)benzaMide Benzamide, N-(2-piperidinylmethyl)-2,5-bis(2,2,2-trifluoroethoxy)- |
| CAS | 54143-55-4 |
| EINECS | 685-650-9 |
| InChI | InChI=1/C17H20F6N2O3/c18-16(19,20)9-27-12-4-5-14(28-10-17(21,22)23)13(7-12)15(26)25-8-11-3-1-2-6-24-11/h4-5,7,11,24H,1-3,6,8-10H2,(H,25,26) |
54143-55-4 - Physico-chemical Properties
| Molecular Formula | C17H20F6N2O3 |
| Molar Mass | 414.34 |
| Density | 1.286±0.06 g/cm3(Predicted) |
| Melting Point | 105-1070C |
| Boling Point | 434.9±45.0 °C(Predicted) |
| Flash Point | 9℃ |
| Solubility | Chloroform (Slightly), Methanol (Slightly) |
| Appearance | Solid |
| Color | White to Off-White |
| pKa | pKa 9.3 (Uncertain) |
| Storage Condition | Keep in dark place,Inert atmosphere,Room temperature |
| Physical and Chemical Properties | UV absorption maximum (ethanol):205,230,300nm(E1 cm1r1, 219,59). Flecainide Monoacetate: C17H20F6N2O3? C2H4O2. [54143-56-5]. White granular crystals were obtained from isopropanol-isopropyl ether, melting point 145~147 °c. Solubility at 37 °c (rng/ml): Water 48.4, ethanol 300. |
54143-55-4 - Risk and Safety
| Risk Codes | R11 - Highly Flammable R23/24/25 - Toxic by inhalation, in contact with skin and if swallowed. R39/23/24/25 - |
| Safety Description | S7 - Keep container tightly closed. S16 - Keep away from sources of ignition. S36/37 - Wear suitable protective clothing and gloves. S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) |
| UN IDs | 3249 |
| WGK Germany | 1 |
| Hazard Class | 6.1(b) |
| Packing Group | III |
54143-55-4 - Reference Information
| NIST chemical information | Information provided by: webbook.nist.gov (external link) |
| uses | new, broad-spectrum and highly effective antiarrhythmic drugs. It is used for the prevention and treatment of a variety of arrhythmia, such as supraventricular tachycardia, atrial fibrillation, single-source and multi-source ventricular premature pulsation syndrome and other cases where antiarrhythmic drugs are ineffective. Can be used as the drug of choice. flucarib is an IC antiarrhythmic drug in Vaughn-Williams classification. it is a cardiac sodium channel blocker and is recommended for cardioversion of atrial fibrillation patients with recent attacks without organic heart disease. Adverse cardiac effects include moderate negative inotropic effects and inhibition of all conduction pathways. |
| Production method | Method 1: Using 2,5-dihydroxybenzoic acid as raw material, after trifluoroethylation, 2-aminomethylpyridine is acylated to obtain amide, and finally fluorocarbini is hydrogenated in the presence of platinum oxide. Method 2: 10.0g(0.249 mo1)2,5-bis (2,2, 2-trifluoroethoxy) benzoic acid -2,2, 2-trifluoroethyl acid was added dropwise to 28.4g(0.0249 mo1) 2-aminomethylpiperidine under nitrogen protection within 25min. After 3 hours, 50ml benzene was added to the viscous reaction solution, and stirring was continued at 45 ℃ for 40 hours. Heat concentration under reduced pressure to remove volatile components. The remainder solidified after cooling, subjected to steam distillation for further purification, filtration, and extraction with dichloromethane. The extract was washed with saturated sodium chloride aqueous solution and dried with anhydrous magnesium sulfate. Filter to remove desiccant, add 4ml of 8.4mol/L isopropanol solution of hydrogen chloride to the filtrate under stirring. After stirring for 2 hours, cool to 0 ℃, filter and collect the crude product, wash with ether, and dry in vacuum. Then the activated carbon was decolorized and recrystallized with the same amount of isopropanol and methanol to obtain fluecainide hydrochloride with a melting point of 228~229 ℃. |
Last Update:2024-04-09 21:11:58
Supplier List
Product Name: Flecainide Request for quotation
CAS: 54143-55-4
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 54143-55-4
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Multiple Specifications
Product Name: Flecainide Visit Supplier Webpage Request for quotationCAS: 54143-55-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: Flecainide Request for quotation
CAS: 54143-55-4
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 54143-55-4
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Multiple Specifications
Product Name: Flecainide Visit Supplier Webpage Request for quotationCAS: 54143-55-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History

