54-16-0
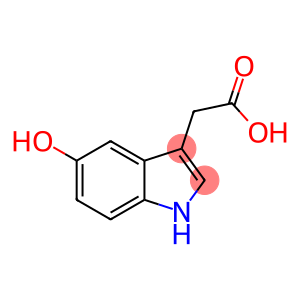
5-Hydroxyindole-3-acetic acid
CAS: 54-16-0
Molecular Formula: C10H9NO3
54-16-0 - Names and Identifiers
| Name | 5-Hydroxyindole-3-acetic acid |
| Synonyms | TIMTEC-BB SBB003639 Hydroxyindoleacetic Acid 5-Hydroxyindolylacetic acid 5-Hydroxyindole-3-acetic acid 5-hydroxyindol-3-ylacetic acid 5-Hydroxy-3-indolyl acetic acid HYDROXYINDOLYL-3-ACETIC ACID, 5- (5-hydroxy-1H-indol-3-yl)acetate 1H-Indole-3-acetic acid, 5-hydroxy- 2-(5-hydroxy-1H-indol-3-yl)acetic acid 5-HYDROXYINDOLE-3-ACETIC ACID FREE ACID |
| CAS | 54-16-0 |
| EINECS | 200-195-4 |
| InChI | InChI=1/C10H9NO3/c12-7-1-2-9-8(4-7)6(5-11-9)3-10(13)14/h1-2,4-5,11-12H,3H2,(H,13,14)/p-1 |
| InChIKey | DUUGKQCEGZLZNO-UHFFFAOYSA-N |
54-16-0 - Physico-chemical Properties
| Molecular Formula | C10H9NO3 |
| Molar Mass | 191.18 |
| Density | 1.2722 (rough estimate) |
| Melting Point | 161-164 °C (dec.) (lit.) |
| Boling Point | 326.92°C (rough estimate) |
| Flash Point | 254.4°C |
| Solubility | Soluble in 50 mg/ml ethanol. |
| Vapor Presure | 1.07E-10mmHg at 25°C |
| Appearance | Light yellow crystal |
| Color | off-white to purple |
| BRN | 168797 |
| pKa | 4.54±0.30(Predicted) |
| Storage Condition | -20°C |
| Sensitive | Light Sensitive |
| Refractive Index | 1.5310 (estimate) |
| MDL | MFCD00005639 |
| In vitro study | 5-Hydroxyindole-3-acetic acid (5HIAA) is a metabolite issued from serotonin metabolism, and the latter is synthetized from the essential amino acid tryptophan. Serotonin is mainly metabolised by monoamine oxidase in the liver and the lungs. |
54-16-0 - Risk and Safety
| Safety Description | S22 - Do not breathe dust. S24/25 - Avoid contact with skin and eyes. |
| WGK Germany | 3 |
| RTECS | NL3650000 |
| FLUKA BRAND F CODES | 8-10-23 |
| HS Code | 29339980 |
54-16-0 - Reference
| Reference Show more | 1. Huang Lin, Cui Ying, Lin, Chen Yai, et al. Effects of Jiawei San Yi decoction on behavioral symptoms and 5-HT 5-HIAA content in Hypothalamus of migraine model rats [J]. Shi Zhen, Chinese medicine 2016(8):1839-1841. 2. Zhu-Hong Zheng, renyun Zhao, Yuting Ding, et al. Study on sedative and hypnotic activities of different extracts from Baihe Dihuang decoction [J]. Northwest Pharmaceutical Journal, 2019, v.34(03):66-70. 3. [IF = 2.952] Chungang Liu et al."Paecilomyces tenuipes extract prevention depression-like behaviors in chronic unpredicted mild stress-induced rat model via modulation of neurotransmitters." Mol Med Rep. 2017 Aug;16(2):2172-2178 4. [IF = 9.028] Feng Cui et al."Toxicological Study on Chiral Fluoxetine Exposure to Adult Zebrafish (Danio rerio): Enantioselective and Sexual Mechanism on Disruption of the Brain Serotonergic System."Environ Sci Technol. 2021;55(11):7479-7490 5. [IF=7.963] Chenzhi Hou et al."Application of multi-parameter population model based on endogenous population biomarkers and flow volume in wastewater epidemiology."Sci Total Environ. 2021 Mar;759:143480 6. [IF=3.932] Yiwen Yang et al."Nyctinastic herbs decoction improves para-chlorophenylalanine-induced insomnia by regulating the expression level of neurotransmitters."Ann Transl Med. 2021 Oct; 9(20): 1524 7. [IF=3.512] Yu Sun et al."Integrated Screening of Effective Anti-Insomnia Fractions of Zhi-Zi-Hou-Po Decoction via Drosophila melanogaster and Network Pharmacology Analysis of the Underlying Pharmacodynamic Material and Mechanism."Acs Omega. 2021;6(13):9176-9187 8. [IF=3.361] Ruixue Yu et al."Targeted neurotransmitter metabolomics profiling of oleanolic acid in the treatment of spontaneously hypertensive rats."Rsc Adv. 2019 Jul;9(40):23276-23288 |
54-16-0 - Reference Information
| Biological activity | 5-Hydroxyindole-3-acetic acid is the main metabolite of serotonin or methoxyepinephrine and is used as a biomarker of neuroendocrine tumors. |
Last Update:2024-04-09 15:16:51
Supplier List
Multiple SpecificationsSpot supply
Product Name: 5-Hydroxyindole-3-acetic Acid Visit Supplier Webpage Request for quotationCAS: 54-16-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 5-HYDROXYINDOLE-3-ACETIC ACID Request for quotation
CAS: 54-16-0
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 54-16-0
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Multiple SpecificationsSpot supply
Product Name: 5-HIAA Visit Supplier Webpage Request for quotationCAS: 54-16-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Multiple SpecificationsSpot supply
Product Name: 5-Hydroxyindole-3-acetic Acid Visit Supplier Webpage Request for quotationCAS: 54-16-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 5-HYDROXYINDOLE-3-ACETIC ACID Request for quotation
CAS: 54-16-0
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 54-16-0
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Multiple SpecificationsSpot supply
Product Name: 5-HIAA Visit Supplier Webpage Request for quotationCAS: 54-16-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


