53544-13-1
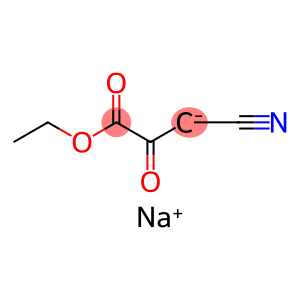
Propanoic acid, 3-cyano-2-oxo-, ethyl ester, ion(1-), sodium
CAS: 53544-13-1
Molecular Formula: C6H6NNaO3
53544-13-1 - Names and Identifiers
53544-13-1 - Physico-chemical Properties
| Molecular Formula | C6H6NNaO3 |
| Molar Mass | 163.11 |
| Storage Condition | under inert gas (nitrogen or Argon) at 2-8°C |
53544-13-1 - Introduction
Propanoic acid, 3-cyano-2-oxo-, ethyl ester, ion(1-), sodium(Propanoic acid, 3-cyano-2-oxo-, ethyl ester, ion(1-), sodium) is an organic compound with the formula C6H5NO2 and a negatively charged sodium ion.
Nature:
-Appearance: Colorless to light yellow liquid
-Density: about 1.23g/cm3
-Melting point: about -112 ℃
-Boiling point: about 222 ℃
-Solubility: Soluble in water and many organic solvents
Use:
Propanoic acid, 3-cyano-2-oxo-, ethyl ester, ion(1-), sodium have many applications in organic synthesis:
-As an intermediate in organic synthesis, used to prepare other organic compounds
-used in the field of medicine, such as synthetic drugs or bioactive molecules
-Ingredients used as fungicides or herbicides in agriculture
Preparation Method:
A commonly used preparation method is to react 3-cyano-2-oxopropionic acid with ethanol under acidic conditions, and then use sodium hydroxide to give a negative charge to generate Propanoic acid, 3-cyano-2-oxo-, ethyl ester, ion(1-), sodium At the same time, sodium ions are formed.
Safety Information:
Propanoic acid, 3-cyano-2-oxo-, ethyl ester, ion(1-), sodium specific hazard information can be provided according to its safety data sheet. Generally speaking, appropriate safety precautions should be taken, such as wearing appropriate personal protective equipment (such as gloves, goggles and protective clothing), and avoiding inhalation, ingestion or contact with skin during handling and storage. In addition, it should be used in a well-ventilated place and away from fire and oxidizing agents.
Nature:
-Appearance: Colorless to light yellow liquid
-Density: about 1.23g/cm3
-Melting point: about -112 ℃
-Boiling point: about 222 ℃
-Solubility: Soluble in water and many organic solvents
Use:
Propanoic acid, 3-cyano-2-oxo-, ethyl ester, ion(1-), sodium have many applications in organic synthesis:
-As an intermediate in organic synthesis, used to prepare other organic compounds
-used in the field of medicine, such as synthetic drugs or bioactive molecules
-Ingredients used as fungicides or herbicides in agriculture
Preparation Method:
A commonly used preparation method is to react 3-cyano-2-oxopropionic acid with ethanol under acidic conditions, and then use sodium hydroxide to give a negative charge to generate Propanoic acid, 3-cyano-2-oxo-, ethyl ester, ion(1-), sodium At the same time, sodium ions are formed.
Safety Information:
Propanoic acid, 3-cyano-2-oxo-, ethyl ester, ion(1-), sodium specific hazard information can be provided according to its safety data sheet. Generally speaking, appropriate safety precautions should be taken, such as wearing appropriate personal protective equipment (such as gloves, goggles and protective clothing), and avoiding inhalation, ingestion or contact with skin during handling and storage. In addition, it should be used in a well-ventilated place and away from fire and oxidizing agents.
Last Update:2024-04-09 20:52:54
Supplier List
Spot supply
Product Name: Sodium 1-cyano-3-ethoxy-2,3-dioxopropan-1-ide Visit Supplier Webpage Request for quotationCAS: 53544-13-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 3-CYANO-2-OXO-PROPANOIC ACID ETHYL ESTER Request for quotation
CAS: 53544-13-1
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 53544-13-1
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Spot supply
Product Name: Sodium 1-cyano-3-ethoxy-2,3-dioxopropan-1-ide Visit Supplier Webpage Request for quotationCAS: 53544-13-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Sodium 1-cyano-3-ethoxy-2,3-dioxopropan-1-ide Visit Supplier Webpage Request for quotationCAS: 53544-13-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 3-CYANO-2-OXO-PROPANOIC ACID ETHYL ESTER Request for quotation
CAS: 53544-13-1
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 53544-13-1
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Spot supply
Product Name: Sodium 1-cyano-3-ethoxy-2,3-dioxopropan-1-ide Visit Supplier Webpage Request for quotationCAS: 53544-13-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


