518-34-3
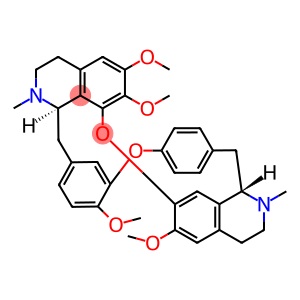
(S,S)-(+)-tetrandrine
CAS: 518-34-3
Molecular Formula: C38H42N2O6
518-34-3 - Names and Identifiers
| Name | (S,S)-(+)-tetrandrine |
| Synonyms | Conba Jinake Trandrine Tetrandrin Tetrandrine D-Tetrandrine (+)-Tetrandrine (S,S)-(+)-tetrandrine TETRANDRINE, (S,S)-(+)-(RG) (1β)-6,6',7,12-tetramethoxy-2,2'-dimethylberbaman 6,6',7,12-Tetramethoxy-2,2'-dimethyl-1-beta-berbaman (1beta)-6,6',7,12-tetramethoxy-2,2'-dimethylberbaman (1beta,1'xi)-6,6',7,12-tetramethoxy-2,2'-dimethylberbaman Berbaman, 6,6',7,12-tetramethoxy-2,2'-dimethyl-, (1beta)- |
| CAS | 518-34-3 |
| EINECS | 683-095-7 |
| InChI | InChI=1/C38H42N2O6/c1-39-15-13-25-20-32(42-4)34-22-28(25)29(39)17-23-7-10-27(11-8-23)45-33-19-24(9-12-31(33)41-3)18-30-36-26(14-16-40(30)2)21-35(43-5)37(44-6)38(36)46-34/h7-12,19-22,29-30H,13-18H2,1-6H3/t29?,30-/m0/s1 |
518-34-3 - Physico-chemical Properties
| Molecular Formula | C38H42N2O6 |
| Molar Mass | 622.75 |
| Density | 1.1528 (rough estimate) |
| Melting Point | 219-222°C(lit.) |
| Boling Point | 662.81°C (rough estimate) |
| Specific Rotation(α) | 285 º (c=1, CHCl3) |
| Flash Point | 175.8°C |
| Solubility | Almost insoluble in water, petroleum ether, soluble in ether and some organic solvents. |
| Vapor Presure | 4.88E-20mmHg at 25°C |
| Appearance | Colorless needle crystal (ether) |
| Color | Off-White |
| Maximum wavelength(λmax) | ['283nm(EtOH)(lit.)'] |
| Merck | 14,9231 |
| pKa | 7.70±0.20(Predicted) |
| Storage Condition | 2-8°C |
| Refractive Index | 1.5300 (estimate) |
| MDL | MFCD08689909 |
| Physical and Chemical Properties | From the Tetrandrine root of the plant |
| Use | Anti-rheumatic and analgesic drugs for the treatment of rheumatism, hypertension, lung cancer and other diseases |
518-34-3 - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| Risk Codes | 36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S22 - Do not breathe dust. S24/25 - Avoid contact with skin and eyes. S36 - Wear suitable protective clothing. S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. |
| WGK Germany | 3 |
| RTECS | XE9350000 |
| HS Code | 29349990 |
518-34-3 - Reference
| Reference Show more | 1. Yang Jingning, Zhang Yan, Wang Min, et al. Protective effect of tetrandrine on multiple organ dysfunction syndrome induced by wasp toxin in rats [J]. Chinese Journal of Industrial Medicine, 2018, 31(05):12-13 83. 2. Guo Xiaoyu, Zhang Xiaoyue, Li Qiyong, etc. Allelopathy of tetrandrine on Microcystis aeruginosa [J]. Journal of Sanming University, 2017, 034(006):57-61. 3. Zhang Keqing, Dai Zhen, Li Yanping. Protective effect of tetrandrine on myocardial I/R in rats by activating Akt/GSK-3b signaling pathway [J]. New Chinese medicine, 2018, 050(009):12-17. 4. Hu, Xi, Zhu, Rong, Tang, Xin et al. Optimization of extraction process of tetrandrine and Tetrandrine from Tetrandrine by response surface methodology [J]. Chinese national and folk medicine 2020 v.29;No.366(01):29-34. 5. He Wanwan and Zhang Jianwei Li Yunjing Kang Tingguo. Correlation between microscopic characteristic index and chemical composition of root of bean [J]. Chinese Journal of Experimental prescriptions, 2017, 23(01):42-46. 6. He Wanwan and Zhang Jianwei Li Yunjing Kang Tingguo. Correlation between powder color and effective components of radix sophorae tonkinensis [J]. Chinese Journal of Experimental prescriptions, 2017, 23(05):57-62. 7. Mengjuan Jiang, Ruoshi Zhang, Yingli Wang, Wenna Jing, Ying Liu, Yan Ma, Bingjun Sun, Menglin Wang, Peizhuo Chen, Hongzhuo Liu, and Zhonggui He,Reduction-sensitive Paclitaxel Prodrug Self-assembled Nanoparticles with Tetrandrine Effectively Promote Synergi 8. Yang, Zhenzhong, et al. "Deciphering bioactive compounds of complex natural products by tandem mass spectral molecular networking combined with an aggregation-induced emission based probe." Journal of Pharmaceutical Analysis (2020).https://doi.org/10.1016/ 9. [IF=5.64] Wu Dousheng et al."Oleanolic Acid Induces the Type III Secretion System of Ralstonia solanacearum."Front Microbiol. 2015 Dec;0:1466 10. [IF=4.939] Mengjuan Jiang et al."Reduction-sensitive Paclitaxel Prodrug Self-assembled Nanoparticles with Tetrandrine Effectively Promote Synergistic Therapy Against Drug-sensitive and Multidrug-resistant Breast Cancer."Mol Pharmaceut. 2017;14(11):3628-3635 11. [IF=4.769] Zhenzhong Yang et al."Deciphering bioactive compounds of complex natural products by tandem mass spectral molecular networking combined with an aggregation-induced emission based probe."J Pharm Anal. 2020 Nov;: 12. [IF=4.411] Dan Liao et al."Tetrandrine Interaction with ABCB1 Reverses Multidrug Resistance in Cancer Cells Through Competition with Anti-Cancer Drugs Followed by Downregulation of ABCB1 Expression."Molecules. 2019 Jan;24(23):4383 13. [IF=3] Qu Lala et al."Phenotypic assessment and ligand screening of ETA/ETB receptors with label-free dynamic mass redistribution assay."N-S Arch Pharmacol. 2020 Jun;393(6):937-950 |
518-34-3 - Reference Information
| overview | tetrandrine (tetrandrine, Tet), also known as tetrandrine, is a bisbenzyl isoquinoline alkaloid extracted from the root tuber of StephamiatetrandraS.Moore, a new type of calcium antagonist, is the main effective ingredient of tetrandrine; it has anti-inflammatory, analgesic, antihypertensive, anti-silicosis, lowering blood sugar, anti-free radical damage, anti-liver fibrosis and other anti-tumor pharmacological effects; it is clinically used for simple silicosis and coal silicosis. It is also used for early mild hypertension, rheumatic pain, arthralgia, neuralgia, etc. It is also used for the treatment of liver cancer, lung cancer, colon cancer, bladder cancer and leukemia. Fig. 1 is the structural formula |
| Han Fangji | Han Fangji is a dried root tuber of the genus StephaniatetrandraS.Mcore of the Fangji family. The root is irregular cylindrical. It is harvested in autumn, washed, peeled and dried until semi-dry, then cut into small pieces, and then dried. The active ingredients of tetrandrine are alkaloids, mainly tetrandrine and tetrandrine. The total content of alkaloids is 1.5% ~ 2.3%, mainly tetrandrine, the content is about 1%, tetrandrine, the content is about 0.5%, and the content is 0.2%. And several other trace alkaloids. |
| pharmacological effects | 1. preventive and therapeutic effects on liver fibrosis Tet can significantly reduce serum type III procollagen, serum and liver HA, and reduce collagen deposition in liver, suggesting that Tet can be used to treat liver fibrosis in chronic liver diseases. 2. Effect on hypoxic pulmonary hypertension In dogs with hypoxic pulmonary hypertension, Tet can significantly reduce the elevated pulmonary artery pressure and pulmonary vascular resistance, and can improve CO and oxygen transport capacity, but has no obvious effect on systemic circulation and blood gas level. Tet mainly inhibits the development of pulmonary hypertension by inhibiting the proliferation of pulmonary arteriole collagen fibers and inhibiting the thickening and myostasis of pulmonary blood vessels caused by hypoxia, which significantly reduces the stenosis of pulmonary arteriole lumen and reduces pulmonary artery pressure. 3. Therapeutic effect on silicosis Based on the preparation of Proα1(Ⅰ) and Proα1(Ⅲ) collagen cDNA probes, the effect of Tet on m RN A of type Ⅰ and Ⅲ collagen genes in silicosis tissue was observed by spot and in situ molecular hybridization. Therefore, it is believed that the accumulation of collagen in silica lung tissue is caused by the enhancement of collagen gene expression caused by quartz dust. TET can directly or indirectly inhibit the transcription of collagen gene, thereby reducing the synthesis of collagen in diseased tissues. 4. Therapeutic effect on hypertension Tet has a significant antihypertensive effect and can significantly reduce the blood lipid peroxide (LPO) and thromboxane (TXB2) levels in hypertensive patients, significantly increase the levels of SOD and prostacyclin (6-keto-PGF1α), and reduce the ratio of TXB2/6-keto-P GF1α. The antihypertensive mechanism of tetrandrine is caused by blocking the voltage-dependent calcium channel of myocardial and vascular smooth muscle and increasing the level of 6-keto-PGF1α. 5. The therapeutic effect of Tet on angina pectoris can significantly reduce the myocardial oxygen consumption index, and there is no significant difference compared with nifedipine. Therefore, Tet is an effective drug for treating angina pectoris, preventing myocardial infarction and reducing myocardial ischemia-reperfusion injury. 6. Therapeutic effect on ophthalmic diseases (1) Therapeutic effect on experimental uveitis: Tet inhibits experimental uveitis. In addition to its anti-inflammatory effect, it is also related to its inhibition of humoral and cellular immune responses. (2) Effect on anterior membrane inflammation: The mechanism of Tet's anti-anterior membrane inflammation may reduce its intracellular. (3) Effect on retinoblastoma: Tet has no direct inhibitory effect on Rb44 cells at low dose (0. 1μg /mL), but it can significantly increase the killing effect of radiation on Rb44 cells. With the increase of drug concentration, the cell killing rate increased, and there was no significant difference before and after radiation administration. Tet mainly increases the effect of radiotherapy by inhibiting the repair of potential lethal injury of cells. 7. Enhance the effect of anticancer drugs. MTT assay was used to detect the toxic effects of daunorubicin (DNR) and homoharringtonine (HHT) on leukemia cells, and to observe the effect of Tet on the cytotoxicity of the two chemotherapeutic drugs. Results Tet can significantly improve the toxic effects of DNR and HHT on drug-resistant cells (K562/AO2, K562/HHT). |
| extraction and separation | 1. extraction of total alkaloids and separation of lipophilic and hydrophilic alkaloids tetrandrine alkaloids, plant raw materials for powder (root). The principle of extraction and separation is high content or main component and dissolution difference, which is called acid water percolation. The separation method is as follows: 2. Low pressure column chromatography separation of tetrandrine A and B under low pressure (0.5~3kg/cm2, generally, 0.3~1.2kg/cm2) is a column chromatography column with silica gel (or alumina) H or G(50~75 μm) as filler for thin layer chromatography with particle diameter between classical column chromatography (100~200 μm) and HPLC(~ 37 μm). Its basic principle is the same as that of HPLC, and its separation effect is also between classical column and HPLC, the column is installed by the decompression dry method, the layer is tightly and evenly, and the distribution of the chromatography zone is concentrated and neat. At the same time, the best separation solvent system for thin layer chromatography can be directly used for low-pressure column chromatography. It is a separation effect. The equipment is simple, easy to operate and fast. Suitable for constant preparative separation of natural products. (1) column loading decompression dry loading method, chromatography column specification: column length 30cm, inner diameter 2cm, total silica gel about 30g (height about 22cm)2.2 mixing sample adding sample taking about 150mg of tetrandrine, adding a small amount of acetone to heat dissolve (just dissolve degree) onto 1.5g silica gel with a dropper, carefully mix well, steam dry in water bath, grind fine, carefully add it to the top of the column through a long neck funnel, and gently and vertically strike, when the surface of the sample is flat and does not move, cover it with blank silica gel about 1 ~ 2cm high, cover it with a circular filter paper sheet, and press it tightly. (2) Elution First check whether the valves and pipelines from the air compressor to the chromatography column are normal, close each valve tightly, and start the air compressor to the rated pressure (5.8kg/cm2) for later use. Carefully add a small amount of eluent (cyclohexane-ethyl ester-diethylamine/6:2:0.8) along the column wall of the chromatography column with a dropper. When the liquid level reaches a certain height, add the rest of the eluent (about 250ml in total) again, quickly install a glass mark plug joint on the top of the column, press it tightly with an iron clamp (the joint is flushed during anti-pressurization), carefully open the air compressor valve, and then open the needle valve and the air filter reducer (note: too much pressure. The glass column will fry, generally 2kg is safe, and a protective mask can be worn when necessary) to mobilize the required pressure, 0.6~1.2kg/cm2, flow out after about 40 minutes, control the flow rate of 1ml/minute, one tube every 10 minutes or so, collect 12~15 parts, and elute the whole process for about 3 hours. (3) Check that each fluid is respectively transferred into a small glass evaporator, concentrated on a water bath, and passed TLC inspection respectively. Adsorbent: silica gel G, developing agent: cyclohexane-ethyl acetate-diethylamine/6:3:1, modified bismuth potassium iodide reagent spray color development, using tetrandrine and ethyl as standard controls, combining the same components to obtain crude A and ethyl respectively, recrystallized with acetone, and determined. |
| physical and chemical identification | take 1g of this product powder, add 15ml of ethanol, heat and reflux for 1 hour, cool, filter, evaporate the filtrate to dry, add 5ml of ethanol to dissolve the residue, and serve as the test solution for the test product. In addition, a reference substance of tetrandrine and tetrandrine is taken, and chloroform is added to prepare a mixed solution containing 1ml in each 1ml as the reference substance test solution. Absorb the above two test solutions 5 μl each, point on the same silica gel G thin layer plate respectively, use chloroform-acetone-methanol (6:1:1) as the developing agent, unfold, dry, and spray to release bismuth iodide potassium test solution. In the reagent chromatography, spots of the same color are displayed at the corresponding position to the control chromatography. |
| content analysis | 1. titration method accurately weigh the appropriate amount of concentrate extracted from the powder, put it into a beaker, dissolve it with a small amount of water, add an appropriate amount of diatomite (Celite535), mix well, dry it at 150 ℃ in powder form, quantitatively transfer it to a filter paper bag, and place it in a sand extractor, reflux extraction with ether-chloroform-ethanol-10% ammonia (28 :8 :2.5:1) mixed solvent until there is no alkaloid reaction in thin layer examination. The extract is dried by solvent in a water bath, an appropriate amount of diethyl ether-chloroform-ethanol (28 :8 :2.5) mixture is added, refluxed and dissolved and transferred to an evaporation dish, 1.5g of medium neutral alumina is added, mixed well, the solvent is carefully removed from the water bath, dried in an oven at 105 ℃ for half an hour, placed at room temperature in a dryer, loaded at the upper end of an alumina column (column diameter 15cm, loaded with 4g alumina), eluted with ethanol, there is no alkaloid spot in the thin layer inspection, the reflux solvent is eluted until it is dry, a small amount of anhydrous slight heat is added to dissolve, 25ml of 0.05mol/l hydrochloric acid solution is accurately added, 2 drops of methyl red-methylene blue mixed indicator are added, 0.05mol/l sodium hydroxide solution is used to drop to the end point, and the same method is used as blank, and the following formula is calculated: alkaloid content (mg)= NaOH(V1-V2)* 622.7*1000/2000V1 = NaOHml number consumed by blank; v2 = NaOHml consumption of samples 2. High performance liquid chromatography accurately weigh 0.1g over 40 mesh samples in a 5ml volumetric flask, add 4ml mobile phase for cold soaking for 12 hours, ultrasonic extraction for 30min, and take 5ml supernatant for injection. Stainless steel column 250mm * 4mmID, filled with gas YWG-C18; The mobile phase is methanol-acetylene-water (3:1:1) containing 0.06% diethylamine, ultrasonic degassing; Temperature 20-25 ℃, flow rate 1.0 ml/min;λ = 242mm; Paper speed 0.25 mm/min. The linear range is between 0.2-1.5 μg. 3. Reverse high performance liquid chromatography takes about 25.0mg of tetrandrine test substance, accurately scales it, places it in a 25ml volumetric flask, dissolves it with mobile phase and dilutes it to scale, shakes it well to obtain 1.00 mg/ml test substance reserve solution. Precisely absorb the reserve liquid of tetrandrine for the test product, use the mobile phase to prepare a solution with a concentration of 10 μg/ml, shake well, take 10 μl and inject it into the liquid chromatograph for determination, and replace the measured peak face value into the regression equation to calculate the concentration to find the sample content. |
| application | 1. immune system disease tetrandrine can obviously inhibit the development of arthritis and its effect is stronger than aspirin, which can be applied to the treatment of rheumatoid arthritis. 2. Cardiovascular diseases tetrandrine not only has calcium antagonism, but also inhibits T-type and L-type calcium channels in ventricular myocytes, and interacts with M receptors to block calcium-activated potassium channels. It has become a treatment for paroxysmal supraventricular tachycardia. 3. Tetrandrine in digestive system plays a certain role in protecting liver cells and resisting liver fibrosis. 4. Ophthalmic diseases tetrandrine is used to treat ocular inflammation, including uveitis, keratitis and retinopathy caused by it. 5. Other tetrandrine has obvious antagonistic effect on acute nephrotoxicity caused by streptomycin, can reduce the apoptosis and proliferation of renal tubular epithelial cells, protect renal function, and have a protective effect on acute ischemia-reperfusion kidney injury. It can significantly reduce serum creatinine and urea nitrogen levels, improve glomerular filtration rate and renal plasma flow, and delay the development of glomerular sclerosis. (2016-02-01) |
| usage and dosage | for silicosis: oral administration, 60~100mg,3 times/d, withdrawal for 6 days and 1 day. 3 months is a course of treatment. For early mild hypertension: oral, 100mg/time, 3 times/d. It can also be used for severe hypertension and hypertensive crisis: intravenous injection, 120~180mg/time, 2 times/d. |
| attention and taboos | may include mild drowsiness, fatigue, nausea, epigastric discomfort, etc. Long-term use can cause facial pigmentation, which can subside after stopping the drug. Intravenous injection can cause local pain. Patients with liver and kidney insufficiency should be used with caution or disabled. During the medication, liver function and electrocardiogram should be checked every 3 months. |
| uses | antirheumatic and analgesic drugs for rheumatism, hypertension, lung cancer, etc. antirheumatic and analgesic drugs, used to treat rheumatism, hypertension, lung cancer and other diseases |
Last Update:2024-04-09 21:11:58
Supplier List
Spot supply
Product Name: D-Tetrandrine Visit Supplier Webpage Request for quotationCAS: 518-34-3
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Spot supply
Product Name: Tetrandrine Visit Supplier Webpage Request for quotationCAS: 518-34-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: D-Tetrandrine Request for quotation
CAS: 518-34-3
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 518-34-3
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Spot supply
Product Name: Stephania Tetrandra extract Tetrandrine Visit Supplier Webpage Request for quotationCAS: 518-34-3
Tel: +86-(0)731-84213302
Email: sales@staherb.cn
Mobile: +86 13875855783
QQ: 484702402
WhatsApp: +86 18374838656
Linkedin: +86 18374838656
Multiple SpecificationsSpot supply
Product Name: Tetrandrine Visit Supplier Webpage Request for quotationCAS: 518-34-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: D-Tetrandrine Visit Supplier Webpage Request for quotationCAS: 518-34-3
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Spot supply
Product Name: Tetrandrine Visit Supplier Webpage Request for quotationCAS: 518-34-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: D-Tetrandrine Request for quotation
CAS: 518-34-3
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 518-34-3
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Spot supply
Product Name: Stephania Tetrandra extract Tetrandrine Visit Supplier Webpage Request for quotationCAS: 518-34-3
Tel: +86-(0)731-84213302
Email: sales@staherb.cn
Mobile: +86 13875855783
QQ: 484702402
WhatsApp: +86 18374838656
Linkedin: +86 18374838656
Multiple SpecificationsSpot supply
Product Name: Tetrandrine Visit Supplier Webpage Request for quotationCAS: 518-34-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History




