503615-07-4
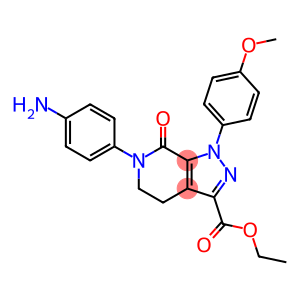
ethyl 6-(4-aminophenyl)-1-(4-methoxyphenyl)-7-oxo-4,5,6,7-tetrahydro-1H-pyrazolo[3,4-c]pyridine-3-carboxylate
CAS: 503615-07-4
Molecular Formula: C22H22N4O4
503615-07-4 - Names and Identifiers
503615-07-4 - Physico-chemical Properties
| Molecular Formula | C22H22N4O4 |
| Molar Mass | 406.43 |
| Density | 1.34±0.1 g/cm3 (20 ºC 760 Torr) |
| Boling Point | 651.6±55.0 °C(Predicted) |
| Solubility | Chloroform (Slightly), DMF (Slightly) |
| Appearance | Solid |
| Color | White to Off-White |
| pKa | 4.66±0.10(Predicted) |
| Storage Condition | Keep in dark place,Inert atmosphere,Store in freezer, under -20°C |
| Use | Intermediate |
503615-07-4 - Reference Information
| apixaban | apixaban (apixaban,1), chemical name is 1-(4-methoxyphenyl) -7-oxo-6-[ 4-(2-oxopiperidine-1-yl) phenyl]-4,5,6, 7-Tetrahydro-1H-pyrazolo [3,4-c] pyridine -3-formamide is an anticoagulant jointly developed by Bristol-Myers Squibb and Pfizer. It directly acts on coagulation factor Xa. It was approved by the European Union and FDA for listing in May 2011 and December 2012 respectively. It was listed in China in April 2013 under the trade name of Eloto. It is used to treat venous thrombotic diseases including deep vein thrombosis and pulmonary embolism . Apixaban is the only new oral anticoagulant approved for secondary prevention at a lower dose than the initial therapeutic dose for VTE. In considering the risks and benefits of continued treatment, patients and physicians may welcome a lower-dose anticoagulant regimen for secondary prevention, and patients are more likely to receive long-term treatment with apixaban, clinical experts note. Studies have shown that apixaban is effective in the treatment of VTE, with fewer bleeding events than warfarin. A new generation of oral anticoagulants gave birth to a new "drug king", which is Pfizer and Bristol-Myers Squibb's apixaban tablets. This third new generation of oral anticoagulants has taken Johnson & Johnson's rivaroxaban tablets to the top of the list. Bristol-Myers Squibb executives recently said in a revenue call that apixaban has become the leader of the new generation of oral anticoagulants based on the overall number of prescriptions. |
| approval status and market status | apixaban was initially approved in the EU in may 2011 (Germany in June, Britain in September, Sweden in December); Approved in the United States in December 2012; Approved in China in January 2013 and Japan in February 2013. For the approved indications, including: (1) treatment of venous thromboembolism (VTE) in adult knee hip replacement surgery (2) prevention of stroke and systemic embolism in adult non-valvular atrial fibrillation (AF) (3) treatment and prevention of deep vein thrombosis (DVT) and pulmonary embolism (PE) for BMS and Pfizer, eliquis is definitely a lucrative drug. Compared with the old anticoagulant warfarin, the Eliquis plays a role through similar blood dilution effect. Patients taking the medicine have fewer bleeding events and good safety, so the market share is increasing year by year. Since its listing in 2011, apixaban's sales have been on the rise. In 2015, global sales exceeded the US $1 billion mark, reaching US $1.86 billion. In 2016, it grew rapidly, with a growth rate of 80% and sales reaching US $3.343 billion. In the first quarter of this year, the overall number of apixaban prescriptions continued to grow by 8%, with a sales performance of 1.1 billion US dollars. This is the first time that the drug has sold more than 1 billion US dollars in a single quarter. According to Thomson Reuters data, its sales will continue to grow in the next five years, with a peak of more than $7 billion! |
| apixaban patent | in 2003, Bristol-Myers Squibb Company of the United States published apixaban compound patent WO2003/049681. patents in China, Europe and the United States correspond to CN1578660A, EP1427415B and US6967208B respectively. The expiration date of the patent is 2022. |
Last Update:2024-04-09 21:01:54
Supplier List
Spot supply
Product Name: 6-(4-Aminophenyl)-1-(4-methoxyphenyl)-7-oxo-4,5,6,7-tetrahydro-1H-pyrazolo[3,4-c]pyridine-3-carboxylic acid ethyl ester Visit Supplier Webpage Request for quotationCAS: 503615-07-4
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: ethyl 6-(4-aminophenyl)-1-(4-methoxyphenyl)-7-oxo-4,5,6,7-tetrahydro-1H-pyrazolo[3,4-c]pyridine-3-carboxylate Visit Supplier Webpage Request for quotationCAS: 503615-07-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: ethyl 6-(4-aMinophenyl)-1-(4-Methoxyphenyl)-7-oxo-4,5,6,7-tetrahydro-1H-pyrazolo[3,4-c]pyridine-3-carboxylate Request for quotation
CAS: 503615-07-4
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 503615-07-4
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: ethyl 6-(4-aminophenyl)-1-(4-methoxyphenyl)-7-oxo-4,5,6,7-tetrahydro-1H-pyrazolo[3,4-c]pyridine-3-carboxylate Visit Supplier Webpage Request for quotation
CAS: 503615-07-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 503615-07-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 6-(4-Aminophenyl)-1-(4-methoxyphenyl)-7-oxo-4,5,6,7-tetrahydro-1H-pyrazolo[3,4-c]pyridine-3-carboxylic acid ethyl ester Visit Supplier Webpage Request for quotationCAS: 503615-07-4
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: ethyl 6-(4-aminophenyl)-1-(4-methoxyphenyl)-7-oxo-4,5,6,7-tetrahydro-1H-pyrazolo[3,4-c]pyridine-3-carboxylate Visit Supplier Webpage Request for quotationCAS: 503615-07-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: ethyl 6-(4-aMinophenyl)-1-(4-Methoxyphenyl)-7-oxo-4,5,6,7-tetrahydro-1H-pyrazolo[3,4-c]pyridine-3-carboxylate Request for quotation
CAS: 503615-07-4
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 503615-07-4
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: ethyl 6-(4-aminophenyl)-1-(4-methoxyphenyl)-7-oxo-4,5,6,7-tetrahydro-1H-pyrazolo[3,4-c]pyridine-3-carboxylate Visit Supplier Webpage Request for quotation
CAS: 503615-07-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 503615-07-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



