500348-26-5
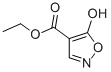
ethyl 5-hydroxyisoxazole-4-carboxylate
CAS: 500348-26-5
Molecular Formula: C6H7NO4
500348-26-5 - Names and Identifiers
500348-26-5 - Physico-chemical Properties
| Molecular Formula | C6H7NO4 |
| Molar Mass | 157.12 |
| Density | 1.328g/cm3 |
| Boling Point | 252.1°C at 760 mmHg |
| Flash Point | 106.2°C |
| Vapor Presure | 0.0124mmHg at 25°C |
| Storage Condition | 2-8℃ |
| Refractive Index | 1.502 |
500348-26-5 - Risk and Safety
| Hazard Class | IRRITANT |
500348-26-5 - Introduction
Ethyl (ethyll) is an organic compound whose structure contains a 5-hydroxyisoxazole ring and a carboxylic acid ethyl ester group.
The following are some of the properties and uses of this compound:
Nature:
-Appearance: Appearance is white crystalline powder.
-Chemical formula: C8H9NO4
-Molecular weight: 179.16g/mol
-Melting point: 85-89°C
-Solubility: Soluble in common organic solvents, such as ethanol and dichloromethane.
Use:
-Chemical synthesis intermediates: Ethyl can be used as drug synthesis intermediates, such as the synthesis of some biologically active compounds.
-Pharmaceutical research: The compound can be used in the field of pharmaceutical research to obtain and study biologically active molecules.
Preparation Method:
ethyl l can generally be synthesized by the following steps:
1. Preparation of 5-hydroxy -4-isoxazole: through appropriate chemical reactions, such as oxidation, acylation and other methods, synthesis of 5-hydroxy -4-isoxazole.
2. reaction: the synthesized 5-hydroxy-4-isoxazole is esterified with ethanolic acid to generate ethyl l.
Safety Information:
- ethyl l may be irritating or harmful to the human body under general conditions, so appropriate operation and protective measures should be taken when using it.
-Avoid contact with ignition sources, oxidants and strong acids during storage and handling to avoid fire or explosion.
-In all cases, the safety data sheets and procedures for chemicals should be read and followed. Users should follow laboratory safety regulations and wear appropriate personal protective equipment, such as lab gloves and protective glasses.
The following are some of the properties and uses of this compound:
Nature:
-Appearance: Appearance is white crystalline powder.
-Chemical formula: C8H9NO4
-Molecular weight: 179.16g/mol
-Melting point: 85-89°C
-Solubility: Soluble in common organic solvents, such as ethanol and dichloromethane.
Use:
-Chemical synthesis intermediates: Ethyl can be used as drug synthesis intermediates, such as the synthesis of some biologically active compounds.
-Pharmaceutical research: The compound can be used in the field of pharmaceutical research to obtain and study biologically active molecules.
Preparation Method:
ethyl l can generally be synthesized by the following steps:
1. Preparation of 5-hydroxy -4-isoxazole: through appropriate chemical reactions, such as oxidation, acylation and other methods, synthesis of 5-hydroxy -4-isoxazole.
2. reaction: the synthesized 5-hydroxy-4-isoxazole is esterified with ethanolic acid to generate ethyl l.
Safety Information:
- ethyl l may be irritating or harmful to the human body under general conditions, so appropriate operation and protective measures should be taken when using it.
-Avoid contact with ignition sources, oxidants and strong acids during storage and handling to avoid fire or explosion.
-In all cases, the safety data sheets and procedures for chemicals should be read and followed. Users should follow laboratory safety regulations and wear appropriate personal protective equipment, such as lab gloves and protective glasses.
Last Update:2024-04-09 20:49:11
Supplier List
Spot supply
Product Name: 5-Hydroxy-4-isoxazolecarboxylic acidethylester Visit Supplier Webpage Request for quotationCAS: 500348-26-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 4-Isoxazolecarboxylic acid, 5-hydroxy-, ethyl ester (9CI) Request for quotation
CAS: 500348-26-5
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 500348-26-5
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: 4-Isoxazolecarboxylic acid, 5-hydroxy-, ethyl ester (9CI) Visit Supplier Webpage Request for quotation
CAS: 500348-26-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 500348-26-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 5-Hydroxy-4-isoxazolecarboxylic acidethylester Visit Supplier Webpage Request for quotationCAS: 500348-26-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 4-Isoxazolecarboxylic acid, 5-hydroxy-, ethyl ester (9CI) Request for quotation
CAS: 500348-26-5
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 500348-26-5
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: 4-Isoxazolecarboxylic acid, 5-hydroxy-, ethyl ester (9CI) Visit Supplier Webpage Request for quotation
CAS: 500348-26-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 500348-26-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


