50-76-0
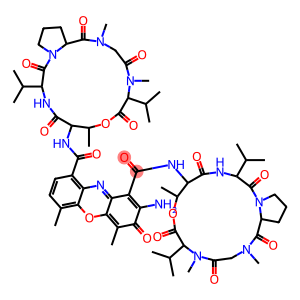
Dactinomycin
CAS: 50-76-0
Molecular Formula: C62H86N12O16
50-76-0 - Names and Identifiers
| Name | Dactinomycin |
| Synonyms | C1 AD X 97 ACTO-D hbf 386 Cosmogen Cosmegen actinomycin7 Oncostatin K Dactinomycin Actinomycin D actinomycin i Actinomycin 7 Meractinomycin dactinomyein d Actinomycin I1 actinomycinaiv actinomycin cl Actinomycin IV Actinomycin C1 Dactinomycin D Actinomycin X 1 actinomycin x i Lyovac cosmegen Actinomycin AIV Dactinomycin, DTM (-)-actinomycin d Actactinomycin A IV Dilactone actinomycin D acid Actinomycin C1, Dactinomycin Dilactone actinomycindioic D acid Actinomycindioic D acid, dilactone actinomyein-theo-val-pro-sar-meval Actinomycin-[threo-val-pro-sar-meval] Actinomycin D, Actinomycin IV, Actinomycin C1, Dactinomycin 2-amino-4,6-dimethyl-3-oxo-N,N'-bis[2,5,9-trimethyl-6,13-bis(1-methylethyl)-1,4,7,11,14-pentaoxooctadecahydrocyclopenta[i][1,4,7,13]oxatriazacyclohexadecin-10-yl]-3H-phenoxazine-1,9-dicarboxamide 2-amino-4,6-dimethyl-3-oxo-N,N'-bis[2,5,9-trimethyl-1,4,7,11,14-pentaoxo-6,13-di(propan-2-yl)hexadecahydro-1H-pyrrolo[2,1-i][1,4,7,10,13]oxatetraazacyclohexadecin-10-yl]-3H-phenoxazine-1,9-dicarboxamide 2-Amino-N,N'-bis[hexadecahydro-2,5,9-trimethyl-6,13-bis(1-methylethyl)-1,4,7,11,14-pentaoxo-1H-pyrrolo[2,1-i][1,4,7,10,13]oxatetraazacyclohexadecin-10-yl]-4,6-dimethyl-3-oxo-3H-phenoxazine-1,9-dicarboxamide 3H-Phenoxazine-1,9-dicarboxamide, 2-amino-N,N'-bis[hexadecahydro-2,5,9-trimethyl-6,13-bis(1-methylethyl)-1,4,7,11,14-pentaoxo-1H-pyrrolo[2,1-i][1,4,7,10,13]oxatetraazacyclohexadecin-10-yl]-4,6-dimethyl-3-oxo- 2-amino-4,6-dimethyl-3-oxo-N,N'-bis[(6S,9R,10S,13R,18aS)-2,5,9-trimethyl-1,4,7,11,14-pentaoxo-6,13-di(propan-2-yl)hexadecahydro-1H-pyrrolo[2,1-i][1,4,7,10,13]oxatetraazacyclohexadecin-10-yl]-3H-phenoxazine-1,9-dicarboxamide |
| CAS | 50-76-0 |
| EINECS | 200-063-6 |
| InChI | InChI=1/C62H86N12O16/c1-27(2)42-59(84)73-23-17-19-36(73)57(82)69(13)25-38(75)71(15)48(29(5)6)61(86)88-33(11)44(55(80)65-42)67-53(78)35-22-21-31(9)51-46(35)64-47-40(41(63)50(77)32(10)52(47)90-51)54(79)68-45-34(12)89-62(87)49(30(7)8)72(16)39(76)26-70(14)58(83)37-20-18-24-74(37)60(85)43(28(3)4)66-56(45)81/h21-22,27-30,33-34,36-37,42-45,48-49H,17-20,23-26,63H2,1-16H3,(H,65,80)(H,66,81)(H,67,78)(H,68,79)/t33-,34-,36+,37+,42-,43-,44+,45+,48+,49+/m1/s1 |
50-76-0 - Physico-chemical Properties
| Molecular Formula | C62H86N12O16 |
| Molar Mass | 1255.42 |
| Density | 1.0757 (rough estimate) |
| Melting Point | 251-253°C |
| Boling Point | 848°C (rough estimate) |
| Flash Point | 87℃ |
| Water Solubility | SOLUBLE |
| Solubility | ethanol, DMSO: Stable in aqueous solutions at 2-8 °C.soluble |
| Vapor Presure | 0mmHg at 25°C |
| Appearance | powder |
| Color | red, powder |
| Merck | 13,2828 |
| BRN | 605235 |
| Storage Condition | 2-8°C |
| Stability | Stable, but light sensitive, especially in dilute solution. Incompatible with strong acids, strong bases, strong oxidizing agents. Combustible. |
| Refractive Index | 1.5700 (estimate) |
| Physical and Chemical Properties | Bright red rhomboid prisms |
50-76-0 - Risk and Safety
| Hazard Symbols | T+ - Very toxic |
| Risk Codes | R28 - Very Toxic if swallowed R61 - May cause harm to the unborn child R40 - Limited evidence of a carcinogenic effect R45 - May cause cancer R26/27/28 - Very toxic by inhalation, in contact with skin and if swallowed. |
| Safety Description | S53 - Avoid exposure - obtain special instructions before use. S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) S1 - Keep locked up. S36/37 - Wear suitable protective clothing and gloves. S28 - After contact with skin, wash immediately with plenty of soap-suds. S22 - Do not breathe dust. |
| UN IDs | UN 3462 6.1/PG 2 |
| WGK Germany | 3 |
| RTECS | AU1575000 |
| FLUKA BRAND F CODES | 8-10-21 |
| HS Code | 29419000 |
| Hazard Class | 6.1(a) |
| Packing Group | II |
| Toxicity | LD50 oral in rat: 7200ug/kg |
50-76-0 - Standard
Authoritative Data Verified Data
This product is actinomycin D. Calculated as dried product, containing actinomycin D(C62H86N12016) shall not be less than 95.0%.
50-76-0 - Trait
Authoritative Data Verified Data
- This product is bright red or deep red crystal, or orange-red crystalline powder; Odorless; Hygroscopicity; Unstable in light.
- This product is soluble in acetone or isopropanol, slightly soluble in methanol, slightly soluble in ethanol, almost insoluble in water, dissolved in 10°C water.
specific rotation
take this product, precision weighing, add methanol to dissolve and quantitatively dilute the solution containing about 1 mg per 1 ml, and determine according to law (General 0621), the specific rotation is from one to 292 ° to one to 317 °.
50-76-0 - Differential diagnosis
Authoritative Data Verified Data
- The solution prepared under the measurement of the content was measured by ultraviolet-visible spectrophotometry (General 0401), and the absorption maximum was found at a wavelength of 241mn and 442nm. The ratio of the absorbance at the wavelength of 241mn to the absorbance at the wavelength of 442mn is 1.3 to 1.5.
- The infrared absorption spectrum of this product should be consistent with that of the control (Spectrum set 177).
50-76-0 - Exam
Authoritative Data Verified Data
crystallinity
take a small amount of this product, according to the law inspection (General 0981), should comply with the provisions.
Related substances
take about 10mg of this product, put it in a 50ml Brown measuring flask, add methanol-water (60:40) to dissolve and dilute to the scale, shake well, as a test solution; take 1ml accurately, put it in a 100ml brown measuring flask, dilute it to the scale with methanol-water (60:40), and shake it well to serve as a control solution. According to high performance liquid chromatography (General rule 0512), octadecylsilane bonded silica gel was used as filler; Acetate buffer solution (sodium acetate 2.72g and acetic acid 2ml were taken, dissolved in water and diluted to ML)-Acetonitrile (49:51) as mobile phase A, with acetate buffer-acetonitrile (20:80) as mobile phase B; Flow rate of 1.5ml per minute, the following table for linear gradient elution; the detection wavelength was 254nm. An appropriate amount of actinomycin D system suitability control was taken, dissolved in methanol-water (60:40) and diluted to make a solution containing about 0.2mg per 1 ml. Take 100u1 injection liquid chromatograph, record the chromatogram should be consistent with the standard map. The retention time of actinomycin D peak is about 30 minutes. Immediately, 100 u1 of the test solution and the control solution are accurately measured, and human liquid chromatograph is injected respectively to record the chromatogram. If there are impurity peaks in the chromatogram of the test solution, the area of a single impurity peak shall not be more than 3 times (3.0%) of the area of the main peak of the control solution, the sum of each impurity peak area shall not be greater than 6 times (6.0%) of the main peak area of the control solution. The impurity peaks in the chromatogram of the test solution which are 0.05 times smaller than the main peak area of the control solution are ignored.
loss on drying
take this product, with phosphorus pentoxide as desiccant, at 60°C under reduced pressure drying to constant weight, weight loss should not exceed 5.0% (General rule 0831).
bacterial endotoxin
take this product, plus endotoxin test water ultrasound to dissolve, according to the law to check (General 1143), each 1 mg of actinomycin D containing endotoxin should be less than 100EU. (For injection)
50-76-0 - Content determination
Authoritative Data Verified Data
take an appropriate amount of this product, accurately weigh, add methanol to dissolve and quantitatively dilute to make a solution containing about 20ug per lml, according to UV-Vis spectrophotometry (General 0401), the absorbance was measured at a wavelength of 442mn and calculated as an absorption coefficient of H86 of 202.
50-76-0 - Category
Authoritative Data Verified Data
Anti-tumor antibiotics.
50-76-0 - Storage
Authoritative Data Verified Data
light-shielded, sealed, and stored in a dry place.
50-76-0 - Actinomycin D for injection
Authoritative Data Verified Data
This product is actinomycin D plus sucrose as excipient made of sterile powder. The content of actinomycin D(C62H86N12O16) shall be between 93.0% and 107.0% of the label weight calculated on average.
trait
This product is light orange-red crystalline powder; Unstable in light.
identification
This product is dissolved in methanol and diluted to prepare a solution containing actinomycin D30ug per 1 ml, filtered and subjected to the identification (1) Test under the item of actinomycin D, showing the same results.
examination
- pH value: Take 1 bottle of this product, add 5ml of water to dissolve it, and measure it according to law (General rule 0631). The pH value should be 5.5~7.5.
- Related substances take 1 bottle of this product, add methanol-water (60:40)lml to dissolve, shake, as a test solution; precision take 100±10ml brown measuring flask, diluted with methanol-water (60:40) to the scale, shake, as a control solution. Take the above two kinds of solution, according to the method of actinomycin D under the determination, for the sample solution chromatogram if there are impurity peaks, single impurity peak area shall not be greater than 3 times (3.0%) of the main peak area of the control solution, the sum of each impurity peak area shall not be greater than 8 times (8.0%) of the main peak area of the control solution.
- weight loss on drying: take this product with phosphorus pentoxide as desiccant, and dry under reduced pressure at 60°C to constant weight. The weight loss shall not exceed 1.0% (General rule 0831). The content uniformity of
- shall be calculated based on the content of each bottle measured under the content determination item, and shall comply with the regulations (General rule 0941).
- bacterial endotoxin should be checked according to the method under actinomycin D, and it should be in accordance with the regulations.
- sterile take this product, dissolve and dilute with appropriate solvent, after the membrane filtration method, inspection according to law (General rule 1101), should comply with the provisions.
- others should comply with the relevant provisions under injection (General 0102).
Content determination
Take 10 bottles of this product, transfer the contents to 10ml measuring flask, add the appropriate amount of methanol, fully shake, dissolve actinomycin D and dilute to the scale, shake, filter, take the filtrate and measure the absorbance at 442nm wavelength by UV-Vis spectrophotometry (General rule 0401). Calculate the content of each bottle according to the absorption coefficient of C62H86N12O16 as 202, the average content of 10 bottles was obtained.
category
isoactinomycin D.
specification
0.2mg
storage
light shielding, closed storage.
50-76-0 - Reference Information
| (IARC) carcinogen classification | 3 (Vol. 10, Sup 7) 1987 |
| EPA chemical substance information | information provided by: ofmpeb.epa.gov (external link) |
| Use | interferon inducer, clinically used as antitumor drug. The product has significant effect on malignant hydatidiform mole, and is effective for nephroblastoma, Hodgkin's disease, testicular tumor, rhabdomyosarcoma, neuroblastoma and reticulocyte sarcoma. Cervical cancer, ovarian cancer, breast cancer and digestive tract cancer, also have a certain effect. Combination with radiotherapy can improve the sensitivity of radiotherapy. curing agent, used for silicone rubber, polyurethane rubber, ethylene-propylene rubber and other rubber; Crosslinking agent, can be used for polyethylene; Unsaturated polyester curing agent. As an effective high-temperature vulcanizing agent for vinyl silicone rubber, polyethylene rubber and ethylene-propylene rubber cross-linking agent can improve the tensile strength and hardness of the product, while the elongation and compression deformation are low, the odor after sulfurization is the most micro in alkyl peroxides |
| production method | This product is a polypeptide antibiotic produced by Actinomycetes. The product extracted from the fermentation broth of Str. Melano chromohes strain isolated from Guilin soil in China is called dactinomycin, which is the same as actinomycin D reported abroad. Using the culture solution of Streptomyces melanogenes as raw material, it was prepared by extraction, acidic alumina column chromatography, reduced pressure concentration and recrystallization from petroleum ether. |
| category | toxic substances |
| toxicity grade | highly toxic |
| Acute toxicity | oral-rat LD50:7.2 mg/kg; Oral-mouse LD50: 13 mg/kg |
| stimulation data | Skin-rabbit 5 mg/24 h positive |
| flammability hazard characteristics | flammability; Combustion releases toxic NOx smoke; Drug side effects: dermatitis, bone marrow injury, digestive system affected |
| storage and transportation characteristics | The warehouse is ventilated and dried at low temperature; Stored separately from food raw materials |
| fire extinguishing agent | dry powder, foam, sand, carbon dioxide, water mist |
| toxic substance data | information provided by: pubchem.ncbi.nlm.nih.gov (external link) |
Supplier List
CAS: 50-76-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
CAS: 50-76-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036

