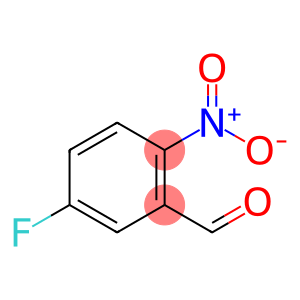
5-fluoro-2-nitrobenzaldehyde
5-fluoro-2-nitrobenzaldehyde
CAS: 395-81-3
Molecular Formula: C7H4FNO3
5-fluoro-2-nitrobenzaldehyde - Names and Identifiers
| Name | 5-fluoro-2-nitrobenzaldehyde |
| Synonyms | 5-Fluoro-2-nitrobenzadehyde 5-FLUORO-2-NITROBENZALDEHYDE 2-NITRO-5-FLUOROBENZALDEHYDE 5-fluoro-2-nitrobenzaldehyde 2-Nitro-5-Fluorobenzaldehyde 4-Fluoro-2-formylnitrobenzene 4-fluoro-2-methoxybenzoic acid Benzaldehyde, 5-fluoro-2-nitro- |
| CAS | 395-81-3 |
| EINECS | 206-903-8 |
| InChI | InChI=1/C8H7FO3/c1-12-7-4-5(9)2-3-6(7)8(10)11/h2-4H,1H3,(H,10,11) |
| InChIKey | KKAFVHUJZPVWND-UHFFFAOYSA-N |
5-fluoro-2-nitrobenzaldehyde - Physico-chemical Properties
| Molecular Formula | C7H4FNO3 |
| Molar Mass | 169.11 |
| Density | 1.443±0.06 g/cm3(Predicted) |
| Melting Point | 92-94°C |
| Boling Point | 153°C 23mm |
| Flash Point | >110°C |
| Water Solubility | Insoluble in water |
| Vapor Presure | 0.00419mmHg at 25°C |
| Appearance | powder to crystal |
| Color | White to Light red to Green |
| BRN | 1869010 |
| Storage Condition | Keep in dark place,Sealed in dry,Room Temperature |
| Sensitive | Air Sensitive |
| Refractive Index | 1.524 |
| Physical and Chemical Properties | 5-fluoro-2-nitrobenzaldehyde is a very light yellow-light red yellow solid at normal temperature and pressure, which is easily oxidized to produce white benzoic acid products and needs to be stored at low temperature. |
5-fluoro-2-nitrobenzaldehyde - Risk and Safety
| Risk Codes | 36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36 - Wear suitable protective clothing. S37 - Wear suitable gloves. |
| HS Code | 29124990 |
| Hazard Class | IRRITANT |
5-fluoro-2-nitrobenzaldehyde - Reference Information
| use | 5-fluoro-2-nitrobenzaldehyde is a synthetic intermediate that can be used as an intermediate in pharmaceutical chemistry and organic synthesis, and can be used to prepare herbicides and plant growth regulators. In the synthetic transformation, the aldehyde group in the structure can be reduced to a hydroxyl group, or react with amine compounds to obtain an imine structure. In addition, the nitro group can be reduced to an amino group, and the obtained amino group can react with the ortho aldehyde group to obtain the imine structure in the molecule. |
| Synthesis method | The mixture of 5-fluoro-2-nitrotoluene (1g, 6.45 mmol) and NBS(2.29g, 12.9 mmol) containing a catalytic amount of AIBN(0.1g, 0.65 mmol) in dry carbon tetrachloride (15 ml) is refluxed for 4 hours, and then the reaction mixture is cooled to room temperature, add another part of NBS(1.15g, 6.45 mmol) and AIBN(0.1g, 0.65 mmol). Then the reaction mixture is refluxed for another 4 hours, cooled to room temperature, and the floating succinimide is filtered out. The resulting filtrate is concentrated under vacuum to obtain a crude bromine compound with a brown viscous liquid. Slowly add tetrabutylammonium dichromate (6.1g, 8.65 mmol) to the solution of the crude bromine compound (1.35g, 5.77 mmol) in dry chloroform (20 ml) and reflux for 2 hours. After the bromine compound is consumed (monitored by TLC), the reaction mixture is concentrated under vacuum, the crude product can be purified by column chromatography (ethyl acetate is 1/99 to petroleum ether) to obtain 5-fluoro-2-nitrobenzaldehyde. Fig. 5-synthesis route of fluoro-2-nitrobenzaldehyde |
Last Update:2024-04-09 21:04:16
Supplier List
Spot supply
Product Name: 5-Fluoro-2-nitrobenzaldehyde Visit Supplier Webpage Request for quotationCAS: 395-81-3
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: 5-Fluoro-2-nitrobenzaldehyde Visit Supplier Webpage Request for quotationCAS: 395-81-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 5-Fluoro-2-nitrobenzadehyde Request for quotation
CAS: 395-81-3
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 395-81-3
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Spot supply
Product Name: 5-Fluoro-2-nitrobenzaldehyde Visit Supplier Webpage Request for quotationCAS: 395-81-3
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: 5-Fluoro-2-nitrobenzaldehyde Visit Supplier Webpage Request for quotationCAS: 395-81-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 5-Fluoro-2-nitrobenzadehyde Request for quotation
CAS: 395-81-3
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 395-81-3
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
View History


