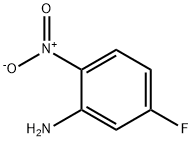
5-Fluoro-2-nitroaniline
5-Fluoro-2-nitroaniline
CAS: 2369-11-1
Molecular Formula: C6H5FN2O2
5-Fluoro-2-nitroaniline - Names and Identifiers
| Name | 5-Fluoro-2-nitroaniline |
| Synonyms | 5-Fluoro-2-nitroanil 5-Fluoro-2-nitroaniline 5-FLUORO-2-NITROANILINE 2-nitro-5-fluoroaniline 5-FLUORO-2-NITROBENZENAMINE 2-Amino-4-fluoronitrobenzene 5-Fluoro-2-nitro-1-benzenamine (5-fluoro-2-nitro-phenyl)amine BenzenaMine, 5-fluoro-2-nitro- |
| CAS | 2369-11-1 |
| EINECS | 219-128-5 |
| InChI | InChI=1/C6H5FN2O2/c7-4-1-2-6(9(10)11)5(8)3-4/h1-3H,8H2 |
| InChIKey | PEDMFCHWOVJDNW-UHFFFAOYSA-N |
5-Fluoro-2-nitroaniline - Physico-chemical Properties
| Molecular Formula | C6H5FN2O2 |
| Molar Mass | 156.11 |
| Density | 1.448±0.06 g/cm3(Predicted) |
| Melting Point | 96.0 to 100.0 °C |
| Boling Point | 295.5±20.0 °C(Predicted) |
| Flash Point | 132.5°C |
| Water Solubility | Slightly soluble in water. |
| Vapor Presure | 0.00152mmHg at 25°C |
| Appearance | Solid |
| pKa | -1.36±0.25(Predicted) |
| Storage Condition | Keep in dark place,Sealed in dry,Room Temperature |
| Refractive Index | 1.603 |
5-Fluoro-2-nitroaniline - Risk and Safety
| Risk Codes | R23/24/25 - Toxic by inhalation, in contact with skin and if swallowed. R33 - Danger of cumulative effects R52/53 - Harmful to aquatic organisms, may cause long-term adverse effects in the aquatic environment. R36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S28 - After contact with skin, wash immediately with plenty of soap-suds. S36/37 - Wear suitable protective clothing and gloves. S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) S61 - Avoid release to the environment. Refer to special instructions / safety data sheets. S24/25 - Avoid contact with skin and eyes. S22 - Do not breathe dust. |
| HS Code | 29214990 |
| Hazard Note | Irritant |
| Hazard Class | 6.1 |
5-Fluoro-2-nitroaniline - Reference Information
| properties | 5-fluoro-2-nitroaniline has a melting point of 96 to 100 degrees, a boiling point of 295.5±20.0 degrees (under one atmospheric pressure), and a solid state under normal temperature and pressure. |
| Use | 5-fluoro-2-nitroaniline can be used as a drug molecule and an intermediate in organic synthesis. Affected by the strong electron withdrawing of the nitro group, the fluorine atom on the benzene ring can be attacked by nucleophiles to obtain derivatized products. For example, under the attack of sodium azide, a product with an azide group at the 5th position of the benzene ring can be obtained. In addition, the nitro group can also be reduced to an amino group under the combination of tin dichloride and hydrochloric acid. |
| synthesis method | 2, 4-difluoronitrobenzene (15 mmol) is dissolved in anhydrous ethanol (10 mL) in a Schlenk flask, adding 25% ammonia solution (10 mL) to the mixture, stirring the reaction mixture at room temperature and ambient temperature, the reaction progress is detected by TLC point plate; after the reaction is over, the precipitate is filtered out by diatomite, the precipitate is washed with water, and the target product 5-fluoro-2-nitroaniline is obtained by drying the precipitate in the air. |
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: 5-Fluoro-2-nitroaniline Visit Supplier Webpage Request for quotationCAS: 2369-11-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: 5-Fluoro-2-nitroaniline Visit Supplier Webpage Request for quotationCAS: 2369-11-1
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: 5-Fluoro-2-nitroaniline Visit Supplier Webpage Request for quotationCAS: 2369-11-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: 5-Fluoro-2-nitroaniline Visit Supplier Webpage Request for quotationCAS: 2369-11-1
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
View History

