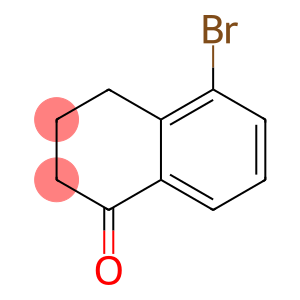
5-Bromo-1-tetralone
5-Bromo-1-tetralone
CAS: 68449-30-9
Molecular Formula: C10H9BrO
5-Bromo-1-tetralone - Names and Identifiers
5-Bromo-1-tetralone - Physico-chemical Properties
| Molecular Formula | C10H9BrO |
| Molar Mass | 225.08 |
| Density | 1.511±0.06 g/cm3(Predicted) |
| Melting Point | 47-48 °C(Solv: methanol (67-56-1)) |
| Boling Point | 105-110 °C(Press: 0.3 Torr) |
| Flash Point | 111.7°C |
| Vapor Presure | 0.000261mmHg at 25°C |
| Storage Condition | Sealed in dry,Room Temperature |
| Refractive Index | 1.598 |
5-Bromo-1-tetralone - Risk and Safety
| Hazard Symbols | Xi - Irritant |
5-Bromo-1-tetralone - Reference Information
| uses | tetralone is an important class of intermediates, widely used in medicine, chemical, new materials and polymer fields, for example, tetralone is an important precursor for the synthesis of antidepressant sertraline, and 6-methoxy-1-naphthone is an important intermediate for the synthesis of family planning drugs such as 18-norgestrel and levonorgestrel, azonaphthone is an important intermediate in the field of new materials and polymers. As one of the tetralone derivatives, 5-bromo-1-tetralone is an important intermediate for the synthesis of selective 5-HT6 receptor antagonists, the antagonist has a potential role in the treatment of central nervous system disorders such as Parkinson's disease and other diseases. |
| preparation | 5-bromo-1-tetralone can be prepared from 5-amino -1-tetralone by the sandemell reaction, however, there is currently no large volume of goods available. The synthetic routes of 5-bromo-1-tetralone can be divided into the following categories:(1) 5-methoxy-1-tetralone is hydrolyzed with hydrobromic acid to give 5-hydroxy-1-tetralone, then it is prepared by alkylation-miles rearrangement-hydrolysis reaction, and this method is not suitable for mass preparation;(2) using benzoyl propionic acid as raw material, after Nitration with mixed acid, after catalytic hydrogenation and reduction of the nitro group, the acetyl group is protected, and the carbonyl group is reduced again, and then the carbonyl group is subjected to Friedel-Crafts acylation to obtain-acetamido-1-tetralone. The total yield of this route is low, and the nitration of mixed acid produces a large amount of waste acid. Therefore, it is necessary to develop a synthetic route with low cost and high yield. In this paper, the cheap and easily available tetrahydronaphthalene is used as raw material to obtain 5-acetyl tetrahydronaphthalene by Friedel-Crafts acylation reaction with acetyl chloride, and the oxime is obtained by hydroxylamine hydrochloride reaction, and then 5-acetyl amino tetrahydronaphthalene is obtained by Beckmann rearrangement, 5-acetamido-1-tetralone was obtained by oxidation, and finally the target product 5-bromo-1-tetralone was obtained by directly carrying out sandemeier reaction through hydrolysis. The raw materials of this route are easily available and easy to operate, suitable for large scale preparation. The synthetic route is shown in Figure 1. Fig.1 reaction formula for synthesis of 5-bromo-1-tetralone |
Last Update:2024-04-09 20:52:54
Supplier List
Spot supply
Product Name: 5-Bromo-3,4-dihydronaphthalen-1(2H)-one Visit Supplier Webpage Request for quotationCAS: 68449-30-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 5-Bromo-1-tetralone Request for quotation
CAS: 68449-30-9
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 68449-30-9
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Product Name: 5-Bromo-1-tetralone Visit Supplier Webpage Request for quotation
CAS: 68449-30-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 68449-30-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 5-Bromo-3,4-dihydronaphthalen-1(2H)-one Visit Supplier Webpage Request for quotationCAS: 68449-30-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 5-Bromo-1-tetralone Request for quotation
CAS: 68449-30-9
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 68449-30-9
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Product Name: 5-Bromo-1-tetralone Visit Supplier Webpage Request for quotation
CAS: 68449-30-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 68449-30-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


