466639-53-2
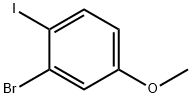
2-bromo-1-iodo-4-methoxy-benzene
CAS: 466639-53-2
Molecular Formula: C7H6BrIO
466639-53-2 - Names and Identifiers
| Name | 2-bromo-1-iodo-4-methoxy-benzene |
| Synonyms | 3-Bromo-4-iodoanisole 2-bromo-1-iodo-4-methoxybenzene 2-BROMO-1-IODO-5-METHOXYBENZENE 2-bromo-1-iodo-4-methoxy-benzene 3-Bromo-4-iodophenyl methyl ether Benzene, 2-bromo-1-iodo-4-methoxy- 2-Bromo-1-iodo-4-methoxybenzene, 3-Bromo-4-iodophenyl methyl ether |
| CAS | 466639-53-2 |
| InChI | InChI=1/C7H6BrIO/c1-10-5-2-3-7(9)6(8)4-5/h2-4H,1H3 |
466639-53-2 - Physico-chemical Properties
| Molecular Formula | C7H6BrIO |
| Molar Mass | 312.93 |
| Density | 2.062±0.06 g/cm3(Predicted) |
| Boling Point | 284.9±25.0 °C(Predicted) |
| Flash Point | 126.085°C |
| Vapor Presure | 0.005mmHg at 25°C |
| Storage Condition | Keep in dark place,Sealed in dry,Room Temperature |
| Sensitive | Irritant |
| Refractive Index | 1.65 |
| MDL | MFCD12547848 |
466639-53-2 - Risk and Safety
| Hazard Class | IRRITANT |
466639-53-2 - Introduction
It is an organic compound with the chemical formula C7H6BrIO. The following is a description of its nature, use, formulation and safety information:
Nature:
-Appearance: It is a solid that can exhibit white to light yellow crystals.
-Melting point: Its melting point range is between 40°C and 45°C.
-Solubility: It has certain solubility in common organic solvents, such as ethanol, dimethylformamide and dichloromethane.
Use:
-is often used in organic synthesis reaction as an important intermediate.
-It can be used to synthesize a variety of organic compounds, such as drugs, dyes and pesticides.
Preparation Method: The preparation method of
-is obtained by reacting anisole with bromine and iodine. Specific steps are as follows:
1. Dissolve anisole in dichloromethane.
2. adding bromine and iodine reaction. Typically, bromine is added first, followed by iodine, while maintaining the temperature of the reaction mixture at room temperature.
3. After completion of the reaction, the product is obtained by filtration or crystallization.
4. Finally, the product is properly purified and dried.
Safety Information:
-is an organic compound that has a certain degree of harmfulness. In the use or handling should pay attention to the following safety matters:
1. Avoid direct contact with skin, eyes and mucous membranes. Wear appropriate protective gloves, goggles and laboratory clothing when used.
2. Operate in a well-ventilated place to avoid inhaling its vapor or dust.
3. Avoid direct sunlight and high temperature during storage, and keep away from flammable materials and oxidants.
4. Waste and its containers must be disposed of in accordance with local regulations.
Please note that this is only a general introduction to Ni, and the specific nature and safety information should be based on reliable experimental data and relevant information. Be sure to follow the correct laboratory procedures and safety instructions when using.
Nature:
-Appearance: It is a solid that can exhibit white to light yellow crystals.
-Melting point: Its melting point range is between 40°C and 45°C.
-Solubility: It has certain solubility in common organic solvents, such as ethanol, dimethylformamide and dichloromethane.
Use:
-is often used in organic synthesis reaction as an important intermediate.
-It can be used to synthesize a variety of organic compounds, such as drugs, dyes and pesticides.
Preparation Method: The preparation method of
-is obtained by reacting anisole with bromine and iodine. Specific steps are as follows:
1. Dissolve anisole in dichloromethane.
2. adding bromine and iodine reaction. Typically, bromine is added first, followed by iodine, while maintaining the temperature of the reaction mixture at room temperature.
3. After completion of the reaction, the product is obtained by filtration or crystallization.
4. Finally, the product is properly purified and dried.
Safety Information:
-is an organic compound that has a certain degree of harmfulness. In the use or handling should pay attention to the following safety matters:
1. Avoid direct contact with skin, eyes and mucous membranes. Wear appropriate protective gloves, goggles and laboratory clothing when used.
2. Operate in a well-ventilated place to avoid inhaling its vapor or dust.
3. Avoid direct sunlight and high temperature during storage, and keep away from flammable materials and oxidants.
4. Waste and its containers must be disposed of in accordance with local regulations.
Please note that this is only a general introduction to Ni, and the specific nature and safety information should be based on reliable experimental data and relevant information. Be sure to follow the correct laboratory procedures and safety instructions when using.
Last Update:2024-04-09 15:17:57
Supplier List
Product Name: 2-bromo-1-iodo-4-methoxybenzene Request for quotation
CAS: 466639-53-2
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 466639-53-2
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
Spot supply
Product Name: 2-Bromo-1-iodo-4-methoxybenzene Visit Supplier Webpage Request for quotationCAS: 466639-53-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-bromo-1-iodo-4-methoxybenzene Request for quotation
CAS: 466639-53-2
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 466639-53-2
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
Spot supply
Product Name: 2-Bromo-1-iodo-4-methoxybenzene Visit Supplier Webpage Request for quotationCAS: 466639-53-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
View History

