4507-57-7
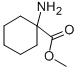
Methyl-1-aminocyclohexane carboxylat
CAS: 4507-57-7
Molecular Formula: C8H15NO2
4507-57-7 - Names and Identifiers
4507-57-7 - Physico-chemical Properties
| Molecular Formula | C8H15NO2 |
| Molar Mass | 157.21 |
| Density | 1.047±0.06 g/cm3(Predicted) |
| Boling Point | 209.9±23.0 °C(Predicted) |
| Flash Point | 79.4°C |
| Vapor Presure | 0.198mmHg at 25°C |
| pKa | 7.19±0.70(Predicted) |
| Storage Condition | 2-8°C(protect from light) |
| Refractive Index | 1.472 |
4507-57-7 - Risk and Safety
| Hazard Class | IRRITANT |
4507-57-7 - Reference Information
| introduction | polyurethane is one of the world's six promising synthetic materials, widely used in plastics, rubber, fibers, coatings and adhesives. Isocyanate is the basic raw material for synthesizing polyurethane. At present, the development of dimethyl carbonate provides a good development opportunity for the green synthesis of isocyanate. The reaction of dimethyl carbonate and organic amine to prepare the corresponding methyl 1-aminocyclohexanarate, and the methyl 1-aminocyclohexanarate is then thermally cracked to form the corresponding isocyanate. This route can effectively avoid the environmental pollution caused by phosgene, which is in line with the trend of chemical industry to clean. |
| Uses | Methyl 1-aminocyclohexanamate is an amino acid compound that can be used as a biochemical reagent. |
| preparation method | methyl 1-aminocyclohexanoic acid was synthesized from methanol and 1-amino -1-cyclohexanoic acid in a yield of about 88%: |
Last Update:2024-04-09 20:45:29
Supplier List
Spot supply
Product Name: Methyl 1-Aminocyclohexanoate Visit Supplier Webpage Request for quotationCAS: 4507-57-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Methyl-1-aminocyclohexane carboxylate (free base) Request for quotation
CAS: 4507-57-7
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 4507-57-7
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
Product Name: Methyl-1-aminocyclohexane carboxylate (free base) Visit Supplier Webpage Request for quotation
CAS: 4507-57-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 4507-57-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Methyl 1-Aminocyclohexanoate Visit Supplier Webpage Request for quotationCAS: 4507-57-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Methyl-1-aminocyclohexane carboxylate (free base) Request for quotation
CAS: 4507-57-7
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 4507-57-7
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
Product Name: Methyl-1-aminocyclohexane carboxylate (free base) Visit Supplier Webpage Request for quotation
CAS: 4507-57-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 4507-57-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


