433-97-6
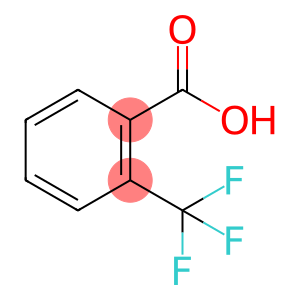
Alpha,Alpha,Alpha-Trifluoro-o-toluic acid
CAS: 433-97-6
Molecular Formula: C8H5F3O2
433-97-6 - Names and Identifiers
| Name | Alpha,Alpha,Alpha-Trifluoro-o-toluic acid |
| Synonyms | ANTIMONY SODIUM FLUORIDE SODIUM ANTIMONY FLUORIDE 2-(Trifluoromethyl)benzoic 2-(trifluoromethyl)benzoate α,α,α-trifluoro-o-toluic acid à,à,à-trifluoro-o-toluic acid 2-trifluoromethylbenzoic acid SODIUM HEXAFLUOROANTIMONATE(V) o-(Trifluoromethyl)benzoic acid 2-(Trifluoromethyl)benzoic acid Alpha,Alpha,Alpha-Trifluoro-o-toluic acid (2Z)-2-chloro-1,1,1,3,4,4,4-heptafluorobut-2-ene |
| CAS | 433-97-6 |
| EINECS | 207-093-9 |
| InChI | InChI=1/C4ClF7/c5-1(3(7,8)9)2(6)4(10,11)12/b2-1- |
| InChIKey | FBRJYBGLCHWYOE-UHFFFAOYSA-N |
433-97-6 - Physico-chemical Properties
| Molecular Formula | C8H5F3O2 |
| Molar Mass | 190.12 |
| Density | 3.375g/mLat 25°C(lit.) |
| Melting Point | 107-110°C(lit.) |
| Boling Point | 247°C753mm Hg(lit.) |
| Flash Point | 247-254°C |
| Water Solubility | 4.8g/L(25 ºC) |
| Vapor Presure | 507mmHg at 25°C |
| Appearance | White solid |
| Color | Slightly yellow to yellow-brown |
| BRN | 976984 |
| pKa | 3.20±0.36(Predicted) |
| Storage Condition | Sealed in dry,Room Temperature |
| Refractive Index | 1.307 |
| MDL | MFCD00002476 |
| Physical and Chemical Properties | O-trifluoromethylbenzoic acid is a white solid, m. P. 109-113 °c, B. P. 247 °c/0.1 MPa, poorly soluble in water, readily soluble in organic solvents. |
433-97-6 - Risk and Safety
| Risk Codes | R20/22 - Harmful by inhalation and if swallowed. R51/53 - Toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment. R36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S61 - Avoid release to the environment. Refer to special instructions / safety data sheets. S36 - Wear suitable protective clothing. S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. |
| UN IDs | UN 1549 6.1/PG 3 |
| WGK Germany | 2 |
| FLUKA BRAND F CODES | 1-10 |
| HS Code | 29163990 |
| Hazard Class | IRRITANT |
433-97-6 - Reference Information
| NIST chemical information | Information provided by: webbook.nist.gov (external link) |
| EPA chemical information | Information provided by: ofmpub.epa.gov (external link) |
| uses | trifluoromethylbenzoic acid compound is a chemical intermediate of medicine and pesticide, which is widely used in medicine, pesticide and other fields. 2-Trifluoromethylbenzoic acid is used in the synthesis of a new broad-spectrum fungicide, flupiramide. The chemical name of the fungicide is: N-{2-[3-chloro -5-(trifluoromethyl) -2-pyridine] ethyl} -2-(trifluoromethyl) benzamide, which can be used for disease control of grapes, pears and stone fruits, vegetables, and field crops. O-trifluoromethylbenzoic acid is an intermediate of the fungicide fluoramide. |
| Preparation | In a 5L stainless steel high-pressure reaction kettle equipped with a mixer and a thermometer, the temperature is reduced to below 5°C, and hydrogen fluoride and 2-trichloromethyldichlorobenzyl are added in sequence, and the mass ratio of the two is 0.2: 1, and then the catalyst perfluorooctane sulfonyl fluoride is added, and the mass ratio of 2-trichloromethyldichlorobenzyl to the catalyst is 1: 0.001, raise the temperature to 80~90 ℃, control the reaction pressure to 2.5-2.8MPa, and react for 4 hours. Sampling, GC detection, the intermediate product 2-difluoro-chloromethyl dichlorobenzyl content is 0.3%. At the end of the reaction, purge with nitrogen to remove excess hydrogen fluoride, and neutralize with potassium carbonate aqueous solution to pH = 6~7. Stand still, separate the product 2-trifluoromethyldichlorobenzyl, the content is 96.6%, the yield is 94.1%. The fluorination reaction product 2-trifluoromethyldichlorobenzyl reacts with nitric acid, and after acidic hydrolysis and oxidation reaction, the target compound 2-trifluoromethylbenzoic acid is obtained. |
| production method | the preparation method is to add 2-chloro-trifluoromethylbenzene to the solvent tetrahydrofuran, then add metal magnesium at 55~60 ℃, add it in batches to cause Grignard reaction, and then add dry ice (carbon dioxide) in ether to stir and react to obtain the finished product. |
Last Update:2024-04-09 21:01:54
Supplier List
Spot supply
Product Name: 2-(Trifluoromethyl)benzoic acid Visit Supplier Webpage Request for quotationCAS: 433-97-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: 2-(trifluoromethyl)benzoic acid Visit Supplier Webpage Request for quotationCAS: 433-97-6
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Product Name: 2-(Trifluoromethyl)benzoic acid Request for quotation
CAS: 433-97-6
Tel: +86-21-56795779
Email: charles7788@worldyachem.com
Mobile: +86-13651600618
QQ: 2850607228
Wechat: 13651600618
WhatsApp: +8613651600618
CAS: 433-97-6
Tel: +86-21-56795779
Email: charles7788@worldyachem.com
Mobile: +86-13651600618
QQ: 2850607228
Wechat: 13651600618
WhatsApp: +8613651600618
Product Name: 2-(Trifluoromethyl)benzoic acid Request for quotation
CAS: 433-97-6
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 433-97-6
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
Spot supply
Product Name: 2-(Trifluoromethyl)benzoic acid Visit Supplier Webpage Request for quotationCAS: 433-97-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 2-(Trifluoromethyl)benzoic acid Visit Supplier Webpage Request for quotationCAS: 433-97-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: 2-(trifluoromethyl)benzoic acid Visit Supplier Webpage Request for quotationCAS: 433-97-6
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Product Name: 2-(Trifluoromethyl)benzoic acid Request for quotation
CAS: 433-97-6
Tel: +86-21-56795779
Email: charles7788@worldyachem.com
Mobile: +86-13651600618
QQ: 2850607228
Wechat: 13651600618
WhatsApp: +8613651600618
CAS: 433-97-6
Tel: +86-21-56795779
Email: charles7788@worldyachem.com
Mobile: +86-13651600618
QQ: 2850607228
Wechat: 13651600618
WhatsApp: +8613651600618
Product Name: 2-(Trifluoromethyl)benzoic acid Request for quotation
CAS: 433-97-6
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 433-97-6
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
Spot supply
Product Name: 2-(Trifluoromethyl)benzoic acid Visit Supplier Webpage Request for quotationCAS: 433-97-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History




