4316-97-6
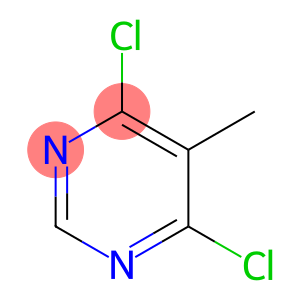
4,6-Dichloro-5-methylpyrimidine
CAS: 4316-97-6
Molecular Formula: C5H4Cl2N2
4316-97-6 - Names and Identifiers
| Name | 4,6-Dichloro-5-methylpyrimidine |
| Synonyms | 4,6-DICHLORO-5-METHYLPYRIMIDIN 4,6-DICHLORO-5-METHYLPYRIMIDINE 4,6-Dichloro-5-methylpyrimidine Pyrimidine, 4,6-dichloro-5-methy 4,6-Dichloro-5-methyl-1,3-diazine Pyrimidine, 4,6-dichloro-5-methyl- 4-chloro-6-methoxy-5-methylpyrimidine |
| CAS | 4316-97-6 |
| InChI | InChI=1/C5H4Cl2N2/c1-3-4(6)8-2-9-5(3)7/h2H,1H3 |
4316-97-6 - Physico-chemical Properties
| Molecular Formula | C5H4Cl2N2 |
| Molar Mass | 163 |
| Density | 1.404g/cm3 |
| Melting Point | 56-60 °C (lit.) |
| Boling Point | 235.399°C at 760 mmHg |
| Flash Point | 118.838°C |
| Solubility | soluble in Methanol |
| Vapor Presure | 0.077mmHg at 25°C |
| Appearance | powder to crystal |
| Color | White to Light yellow to Light orange |
| Storage Condition | Inert atmosphere,2-8°C |
| Refractive Index | 1.551 |
| MDL | MFCD01646112 |
4316-97-6 - Risk and Safety
| Hazard Symbols | C - Corrosive |
| Risk Codes | 34 - Causes burns |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) |
| UN IDs | UN 3261 8/PG 2 |
| WGK Germany | 3 |
| HS Code | 29335990 |
| Hazard Class | CORROSIVE |
4316-97-6 - Reference Information
| Application | 4, 6-dichloro-5-methylpyrimidine is a heterocyclic organic compound, which can be used as an intermediate in organic synthesis. |
| preparation method | the first step is to quickly mix 18.9g of methanol solution 28% sodium methoxide with 4.7g of formamide, and heat the mixture under reflux, and slowly drop 30ml of methanol solution containing 5g of ethyl methylmalonate within 3 hours. The mixture is further heated and refluxed with stirring for 10 hours, and then cooled to concentrate the suspension under reduced pressure. Then, add 10ml of water to the residue and acidify it with concentrated hydrochloric acid. The precipitate obtained was filtered by suction and dried under reduced pressure to obtain 4, 6-dihydroxy-5-methylpyrimidine. 4, 6-dihydroxy-5-methylpyrimidine, 2.8g. In the second step, 2.8g of 4, 6-dihydroxy-5-ethylpyrimidine, 5.00g of phosphorus trichloride and 3.3g of diisopropylethylamine were added to the reaction vessel at 0°C, and then stirred at 80°C for 2 hours. 4 hours. The mixture is then cooled and poured into ice water. The mixture was further stirred for 30 minutes and then extracted with ethyl acetate. The organic layers are combined and washed successively with saturated sodium bicarbonate aqueous solution and saturated sodium chloride aqueous solution, and then the organic layers are dried and concentrated with anhydrous magnesium sulfate. The residue was subjected to silica gel column chromatography to obtain 2.4g of 4, 6-dichloro-5-methylpyrimidine. |
Last Update:2024-04-10 22:29:15
Supplier List
Product Name: 4,6-Dichloro-5-methylpyrimidine Request for quotation
CAS: 4316-97-6
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 4316-97-6
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
Spot supply
Product Name: 4,6-Dichloro-5-methylpyrimidine Visit Supplier Webpage Request for quotationCAS: 4316-97-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 4,6-Dichloro-5-methylpyrimidine Visit Supplier Webpage Request for quotation
CAS: 4316-97-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 4316-97-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: 4,6-Dichloro-5-methylpyrimidine Request for quotation
CAS: 4316-97-6
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 4316-97-6
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
Spot supply
Product Name: 4,6-Dichloro-5-methylpyrimidine Visit Supplier Webpage Request for quotationCAS: 4316-97-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 4,6-Dichloro-5-methylpyrimidine Visit Supplier Webpage Request for quotation
CAS: 4316-97-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 4316-97-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


