41198-02-1
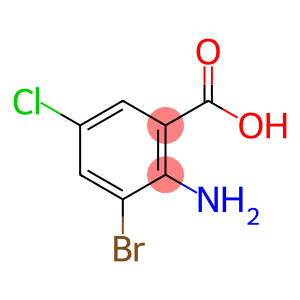
2-Amino-3-bromo-5-chlorobenzoic acid
CAS: 41198-02-1
Molecular Formula: C7H5BrClNO2
41198-02-1 - Names and Identifiers
41198-02-1 - Physico-chemical Properties
| Molecular Formula | C7H5BrClNO2 |
| Molar Mass | 250.48 |
| Storage Condition | Keep in dark place,Sealed in dry,Room Temperature |
41198-02-1 - Introduction
Acid is an organic compound with the chemical formula C7H5BrClNO2. The following is an introduction to some of its properties, uses, methods and safety information:
Nature:
-Appearance: acid is a white crystalline solid.
-Melting point: about 200-202°C.
-Solubility: Its solubility in water is relatively low, and its solubility in organic solvents (such as ethanol and acetone) is better.
Use:
-Chemical synthesis: acid can be used as an intermediate in organic synthesis and used to synthesize other complex compounds.
-Drug research: It can also be applied to the field of drug research as a skeleton of drug molecules.
Preparation Method:
The preparation method of acid usually involves the following steps:
1. First, benzyl p-formate is obtained from benzoic acid by suspension and solvent extraction.
2. Next, benzyl p-formate is reacted with ammonia water to produce p-aminobenzoic acid.
3. Finally, p-aminobenzoic acid reacts with a mixed solution of bromine and chlorine to obtain acid.
Safety Information:
-the acid currently has no public specific safety data, so general laboratory safety regulations and personal protective measures should be followed when using it.
-In case of contact with skin or eyes, rinse immediately with plenty of water and seek the help of a doctor.
-Avoid inhaling the dust or gas of the compound, and operate in a well-ventilated area.
-During use or storage, avoid contact with strong oxidants to avoid dangerous reactions.
-Detailed safety information is available through the chemical provider or the relevant database.
Nature:
-Appearance: acid is a white crystalline solid.
-Melting point: about 200-202°C.
-Solubility: Its solubility in water is relatively low, and its solubility in organic solvents (such as ethanol and acetone) is better.
Use:
-Chemical synthesis: acid can be used as an intermediate in organic synthesis and used to synthesize other complex compounds.
-Drug research: It can also be applied to the field of drug research as a skeleton of drug molecules.
Preparation Method:
The preparation method of acid usually involves the following steps:
1. First, benzyl p-formate is obtained from benzoic acid by suspension and solvent extraction.
2. Next, benzyl p-formate is reacted with ammonia water to produce p-aminobenzoic acid.
3. Finally, p-aminobenzoic acid reacts with a mixed solution of bromine and chlorine to obtain acid.
Safety Information:
-the acid currently has no public specific safety data, so general laboratory safety regulations and personal protective measures should be followed when using it.
-In case of contact with skin or eyes, rinse immediately with plenty of water and seek the help of a doctor.
-Avoid inhaling the dust or gas of the compound, and operate in a well-ventilated area.
-During use or storage, avoid contact with strong oxidants to avoid dangerous reactions.
-Detailed safety information is available through the chemical provider or the relevant database.
Last Update:2024-04-09 21:01:54
Supplier List
Product Name: Benzoic acid, 2-aMino-3-broMo-5-chloro- Request for quotation
CAS: 41198-02-1
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 41198-02-1
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
Spot supply
Product Name: 2-Amino-3-bromo-5-chlorobenzoic acid Visit Supplier Webpage Request for quotationCAS: 41198-02-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-Amino-3-bromo-5-chlorobenzoic Acid Visit Supplier Webpage Request for quotation
CAS: 41198-02-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 41198-02-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: Benzoic acid, 2-aMino-3-broMo-5-chloro- Request for quotation
CAS: 41198-02-1
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 41198-02-1
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
Spot supply
Product Name: 2-Amino-3-bromo-5-chlorobenzoic acid Visit Supplier Webpage Request for quotationCAS: 41198-02-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-Amino-3-bromo-5-chlorobenzoic Acid Visit Supplier Webpage Request for quotation
CAS: 41198-02-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 41198-02-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


