41059-79-4
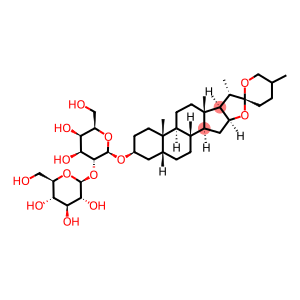
timosaponin A-III
CAS: 41059-79-4
Molecular Formula: C39H64O13
41059-79-4 - Names and Identifiers
| Name | timosaponin A-III |
| Synonyms | Filiferin B XilingsaponinA TIMOSAPONIN A- TIMOSAPONIN A-3 AnemarsaponinA3 Timosaponin A-III timosaponin A-III TIMOSAPONIN A3 (SYN. TIMOSAPONIN A-III) β-D-Galactopyranoside, (3β,5β,25S)-spirostan-3-yl2-O-β-D-glucopyranosyl- |
| CAS | 41059-79-4 |
| InChI | InChI=1/C39H64O13/c1-18-7-12-39(47-17-18)19(2)28-25(52-39)14-24-22-6-5-20-13-21(8-10-37(20,3)23(22)9-11-38(24,28)4)48-36-34(32(45)30(43)27(16-41)50-36)51-35-33(46)31(44)29(42)26(15-40)49-35/h18-36,40-46H,5-17H2,1-4H3/t18?,19-,20+,21-,22+,23-,24-,25-,26+,27+,28-,29+,30-,31-,32-,33+,34+,35-,36+,37-,38-,39+/m0/s1 |
41059-79-4 - Physico-chemical Properties
| Molecular Formula | C39H64O13 |
| Molar Mass | 740.92 |
| Density | 1.36±0.1 g/cm3(Predicted) |
| Melting Point | 317-322 °C |
| Boling Point | 862.8±65.0 °C(Predicted) |
| Solubility | Soluble in methanol, chloroform methanol mixed solvent, hardly soluble in ethyl acetate, chloroform |
| Appearance | Colorless needle crystal |
| pKa | 12.85±0.70(Predicted) |
| Storage Condition | 2-8℃ |
| Refractive Index | 1.606 |
| MDL | MFCD01631164 |
| Physical and Chemical Properties | Soluble in methanol, chloroform methanol mixed solvent, insoluble in ethyl acetate, chloroform from the monocotyledonous liliae Anemarrhena asphodelodides Bge. |
41059-79-4 - Reference
| Reference Show more | 1. Liu Yanping, Yang Yishun, Zhang Tong, etc. Optimization of enzymatic hydrolysis process of timosaponin A Ⅲ [J]. Chinese Journal of Experimental prescriptions, 2013, 19(2):1-4. 2. Wang Xiaoting, Yan Li, Sun lunar November, etc. Effect of incremental components on glucose consumption of HepG-2 cells after preparation of anemarrhena asphotica Bunge [J]. Chinese Journal of Hospital Pharmacy, 2018, v.38(16):29-31. 3. Ni, Wendi, Zhu, Min, Zhou, Qinmei. Determination of two anemarrhena saponins in zishenguanping capsules by HPLC-ELSD [J]. Jiangsu Traditional Chinese Medicine 2012(11):69-70. 4. Zhang Shengan, Tao Chunxiao, Lu Lu, Jiang Yan, Zhou Guoliang, Ding Yue. Effect of enzymatic hydrolysis technology on antifungal activity of Rhizoma anemarrhenae [J]. Journal of Shanghai University of Traditional Chinese Medicine, 2019,33(05):67-72. 5. Ma Ninghui, Lu Lu, Ding Yue, et al. Preparation of doxorubicin hydrochloride and timosaponin A Ⅲ co-loaded liposomes and determination of entrapment efficiency [J]. Chinese herbal medicine, 2019, 50(01):69-75. 6. Liu Y, Pu Y, Zhang T, Ding Y, Wang B, Cai Z. Rapid and Sensitive Determination of Timosaponin AIII in Rat Plasma by LC-MS/MS and Its Pharmacokinetic Application. International Journal of Molecular Sciences. 2013; 14(2):3656-3670. https://doi.org/10.3390/ij 7. [IF=5.923] Yanping Liu et al."Rapid and Sensitive Determination of Timosaponin AIII in Rat Plasma by LC-MS/MS and Its Pharmacokinetic Application."Int J Mol Sci. 2013 Feb;14(2):3656-3670 8. [IF=3.041] Hong Wang et al."Timosaponin A‑III induces autophagy of T‑cell acute lymphoblastic leukemia Jurkat cells via inhibition of the PI3K/Akt/mTOR pathway."Oncol Rep. 2019 May;41(5):2937-2944 9. [IF=2.289] Zhou Shun et al."Anthelmintic efficacy of natural saponins against Gyrodactylus kobayashii in goldfish (Carassius auratus) and their 3D-QSAR analysis."Parasitol Res. 2021 Mar;120(3):1143-1150 |
41059-79-4 - Reference Information
| plant source | anemarrhena is the dry rhizome of the Lilies plant Anemarrhena (Anemarrhena asphodeloides Bge.). It has bitter taste, cold nature, and returns to the lungs, stomach, and kidney meridian. It has the effects of clearing heat and purging fire, nourishing yin and moistening dryness, quenching thirst and eliminating irritability, it is clinically used for symptoms such as exogenous fever, high fever and polydipsia, lung heat and dry cough, bone steaming hot flashes, internal heat and thirst, intestinal dryness and constipation. As the main pharmacological active component of Anemarrhena asphodeloides, the content of asphodeloides in the rhizome of Anemarrhena asphodeloides is about 6%. Anemarrhena saponin A- III(Timosaponin A- Ⅲ), also known as siling saponin A(Xilingsaponin A), is a typical spirosteroidal saponin component, and its parent core is sarsasapogenin [also known as salsa sapogenin (Sarsasapogenin)], is a polyglycoside formed by dehydrogenation of the hydroxyl group at the C22 position of sarsasapogenin with two fructose and one glucose. Anemarrhena AⅢ has anti-platelet aggregation, anti-senile dementia, anti-tumor and hypoglycemic effects. Especially in anti-tumor, it shows strong pharmacological activity, and has inhibitory effects on colon cancer, cervical cancer, breast cancer, etc. |
| chemical composition | rhizome contains about 6% steroidal saponins, including anemarrhena asphosenoside A1, A2, A3, A4, B1, B2, moelcor saponins, etc. The sapogenins of Anemarrhena mainly include sarsasapogenin, marcosidin (Markogenin) and neogitorin (Neogitogenin). fig. 1 is the structural formula of anemarrhena saponin A3 and anemarrhena saponin B1. |
| extraction and separation | 1. extraction and separation of total saponins. 2. extraction and separation of sarsasapogenin, anemarrhena saponin A3, sarsasaponin, sarsasapogenin, anemarrhena saponin B1, anemarrhena saponin B3. 3. extraction and separation of anemarrhena saponin I and anemarrhena saponin I a. 4. extraction and separation of anemarrhena saponin d. 5. extraction and separation of anemarrhena saponin f and anemarrhena saponin g. 6. extraction and separation of anemarrhena saponin E1 and anemarrhena saponin E2. 7. extraction and separation of anemarrhena saponin H1, anemarrhena saponin H2, anemarrhena saponin Ⅰ 1 and anemarrhena saponin Ⅰ 2. 8. extraction and separation of mangiferin and neomangiferin. 9. separation and extraction of polysaccharide. Figure 2 shows the components of Anemarrhena. |
| Pharmacological effects | 1. Pharmacological effects of the cardiovascular system 1.1 anti-platelet aggregation The molecular mechanisms of anti-platelet aggregation A- III by Anemarrhizin are mainly as follows: ① It can activate the platelet P2Y1 receptor coupled to the Gq subunit of the G protein-coupled receptor, thereby inducing platelet deformation; ② it can inhibit Gq and G12/G13 subunits coupled to g protein and prevent them from transmitting activation signals, thus inhibiting platelet aggregation in a dose-dependent manner. ③ can activate Gi signaling pathway, inhibit adenylate cyclase and increase cAMP, thus inhibiting platelet aggregation. ④ can indeed inhibit the binding of platelets and fibrinogen by inhibiting the activation of integrin αⅡbβ3, so as to inhibit the further transmission of the aggregation signal. 1.2 Vasodilation Stimulation of Ca2 + influx can stimulate vascular endothelial cell vasodilator (NO) synthase, and then promote endothelial cell release of NO,NO can in turn stimulate Ca2 + to transfer to endothelial cell, which makes the vasodilator activity of anemoside AⅢ stronger. 2. improving memory and learning ability Bomi et al. found that the A- III of 40 mg/kg of anemarrhena saponin can significantly reduce the number of errors in passive avoidance experiment of glutamate-induced senile dementia model mice, and significantly shorten the latency of errors. Its mechanism of action may be to increase the concentration of norepinephrine, dopamine and serotonin in the brain of dementia mice, thereby improving the memory of mice. At the same time, the A- III of anemarrhena saponin can reduce the number of β-APP positive neurons in the back hippocampus and dentate gyrus of dementia mice, thus improving the symptoms of senile dementia in mice. 3. hypoglycemic effect anemarrhena saponin A- III has the effect of reducing hyperglycemia and its intensity is concentration-dependent. |
| anti-tumor effect | 1, inducing apoptosis of cervical cancer cells (HeLa) the process of self-dissolution of HeLa cells induced by anemarrhena AⅢ will produce a large number of acidic giant autogenous phagocytic bubbles, which are quite different from the small and medium-sized autophagocytic bubbles formed by the traditional active ingredients that induce autolysis of cancer cells, this suggests that this product may have a more powerful effect on inducing apoptosis than traditional anticancer drugs. 2. Anti-colon cancer effect The mechanism of its anti-tumor effect is that it can effectively prevent the cell cycle from going on, and its specific effects are as follows: ① It can effectively reduce the cell quiescent phase (S phase) and DNA mitotic phase (G2 phase) Proto-oncogene C and cell cycle regulatory protein (CDK4) expression and can prevent tumor cell suppressor protein phosphorylation; ② in the cell mitotic preparation phase (G1 phase) and mitotic phase (m phase), anemoside A Ⅲ can significantly down-regulate the expression level of cell regulatory proteins A and B1. 3. Anti-breast cancer effect Anemoside AⅢ induces apoptosis mainly through activation of endoplasmic reticulum corposome stress. |
| use | used for content determination/identification/pharmacological experiments, etc. Pharmacological effects: After acting on vascular endothelial cells, the expression of endothelial converting enzyme gene will be down-regulated to varying degrees, which can regulate the function of vascular endothelial cells. |
Last Update:2024-04-09 15:16:50
Supplier List
Spot supply
Product Name: Timosaponin A3 Visit Supplier Webpage Request for quotationCAS: 41059-79-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Timosaponin A-III Request for quotation
CAS: 41059-79-4
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 41059-79-4
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
Spot supply
Product Name: Timosaponin A3 Visit Supplier Webpage Request for quotationCAS: 41059-79-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Timosaponin A3 Visit Supplier Webpage Request for quotationCAS: 41059-79-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Timosaponin A-III Request for quotation
CAS: 41059-79-4
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 41059-79-4
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
Spot supply
Product Name: Timosaponin A3 Visit Supplier Webpage Request for quotationCAS: 41059-79-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


