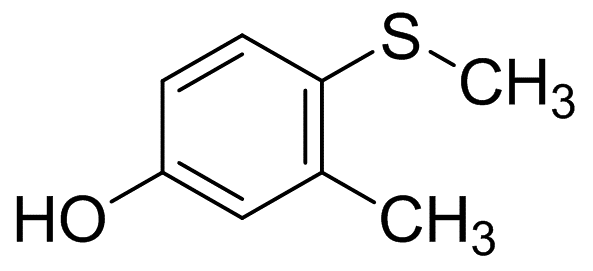
4-Methylthio-M-Cresol
4-Methylthio-M-Cresol
CAS: 3120-74-9
Molecular Formula: C8H10OS
4-Methylthio-M-Cresol - Names and Identifiers
| Name | 4-Methylthio-M-Cresol |
| Synonyms | MTMC mmtp usafma-17 Methylthiomcresol 4-Methylthio-M-Cresol 4-(methylthio)-m-creso Methylthiomethylphenol 4-(Methylthio)-m-cresol 3-methyl-4-sulfanylphenol P-METHYLMERCAPTO-M-CRESOL 3-Methyl-4-(methylthio)phenol 3-methyl-4-(methylthio)-pheno 3-Methyl-4-methylsulfanylphenol 3-methyl-4-(methylsulfanyl)phenol 4-methoxy-3-(methylsulfanyl)phenol |
| CAS | 3120-74-9 |
| EINECS | 221-496-7 |
| InChI | InChI=1/C8H10O2S/c1-10-7-4-3-6(9)5-8(7)11-2/h3-5,9H,1-2H3 |
4-Methylthio-M-Cresol - Physico-chemical Properties
| Molecular Formula | C8H10OS |
| Molar Mass | 154.23 |
| Density | 1.1125 (rough estimate) |
| Melting Point | 56-60 °C |
| Boling Point | 151 °C / 15mmHg |
| Flash Point | 137.191°C |
| Solubility | Chloroform, Methanol |
| Vapor Presure | 0.001mmHg at 25°C |
| Appearance | Solid |
| Color | Off-White to Light Beige |
| BRN | 1210322 |
| pKa | 9.90±0.18(Predicted) |
| Storage Condition | Room Temperature |
| Refractive Index | 1.5300 (estimate) |
| MDL | MFCD00045773 |
| Physical and Chemical Properties | p-methylthio m-cresol is colorless needle crystal, m.p.56 ~ 57 ℃, B. p.186 ℃/2.67kPa, relative density 1.10, insoluble in water, soluble in organic solvent. |
| Use | Used as an intermediate in organic synthesis |
4-Methylthio-M-Cresol - Risk and Safety
| Risk Codes | R22 - Harmful if swallowed R37/38 - Irritating to respiratory system and skin. R41 - Risk of serious damage to eyes |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36/39 - |
| UN IDs | 3335 |
| WGK Germany | 3 |
| RTECS | GP2225000 |
| TSCA | T |
| Hazard Class | IRRITANT, STENCH |
4-Methylthio-M-Cresol - Nature
Open Data Verified Data
light red crystal, melting point 52~54 ℃, boiling point 224~228 ℃. Insoluble in water, soluble in methanol, ethanol, easy oxidation in the air, a special odor.
Last Update:2024-01-02 23:10:35
4-Methylthio-M-Cresol - Preparation Method
Open Data Verified Data
prepared by the reaction of m-cresol with dimethyl disulfide.
Last Update:2022-01-01 10:56:04
4-Methylthio-M-Cresol - Use
Open Data Verified Data
This product can be used for the synthesis of organophosphorus insecticide, and can also be used for the synthesis of nematicide.
Last Update:2022-01-01 10:56:03
4-Methylthio-M-Cresol - Safety
Open Data Verified Data
25kg/paper drum.
Last Update:2022-01-01 10:56:04
4-Methylthio-M-Cresol - Reference Information
| chemical properties | p-methylthio m-cresol is colorless needle crystal, m.p.56 ~ 57 ℃, B. p.186 ℃/2.67kPa, relative density 1.10, insoluble in water, soluble in organic solvent. |
| use | p-methylthio-cresol, I .e. 3-methyl -4-methylthio phenol, is used to synthesize insecticides fenthion, aniline fenthion and nematicide fenthion. Pesticide intermediates. Used as an intermediate in organic synthesis |
| Production method | It is obtained by the reaction of m-cresol and dimethyl disulfide. Mix m-cresol and dimethyl disulfide, add 95% sulfuric acid dropwise, control the reaction temperature to 10-15 ℃, separate waste acid after 5 hours of reaction, neutralize with soda ash solution, separate water layer, wash with water, and evaporate under reduced pressure to remove water, unreacted dimethyl disulfide and cresol to obtain 3-methyl -4-methylthiophenol. the preparation of p-methylthio m-cresol mainly includes dimethyl disulfide method and dimethyl sulfoxide method. (1) The dimethyl disulfide method is obtained by the reaction of m-cresol and dimethyl disulfide. Add m-cresol and dimethyl disulfide into the reaction kettle, stir, cool to 0 ℃, add sulfuric acid dropwise, add it for about 1h, keep the temperature below 5 ℃ for 3h, and let it stand for stratification. The organic layer is neutralized with 10% sodium carbonate solution, then washed with water to pH = 7~8, distilled under reduced pressure, and unreacted m-cresol and dimethyl disulfide are recovered, and the remaining material is p-methylthio-m-cresol. (2) The dimethyl sulfoxide method uses m-cresol and dimethyl sulfoxide as raw materials. This method is divided into hydrogen chloride method, phosgene method and benzoyl chloride method. The hydrogen chloride method is mainly used in China. Mix dimethyl sulfoxide and m-cresol and stir, and pass hydrogen chloride gas at a certain temperature until it is saturated, until a large amount of white smoke escapes. Add a certain amount of m-cresol as a decomposition solvent (usually xylene can also be used) to the white pasty sulfonium salt generated by the reaction, stir for several minutes at room temperature, drive out the hydrogen chloride dissolved in the sulfonium salt, and then reflux at 120~130 ℃ for 1.5~2h, and evaporate m-cresol under reduced pressure to obtain the product. |
| EPA chemical information | information provided by: ofmpub.epa.gov (external link) |
Last Update:2024-04-09 20:52:54
Supplier List
Spot supply
Product Name: 3-Methyl-4-(methylthio)phenol Visit Supplier Webpage Request for quotationCAS: 3120-74-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 3-Methyl-4-(methylthio)phenol Request for quotation
CAS: 3120-74-9
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 3120-74-9
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: 3-Methyl-4-(Methylthio)Phenol Visit Supplier Webpage Request for quotation
CAS: 3120-74-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 3120-74-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 3-Methyl-4-(methylthio)phenol Visit Supplier Webpage Request for quotationCAS: 3120-74-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 3-Methyl-4-(methylthio)phenol Request for quotation
CAS: 3120-74-9
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 3120-74-9
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: 3-Methyl-4-(Methylthio)Phenol Visit Supplier Webpage Request for quotation
CAS: 3120-74-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 3120-74-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


