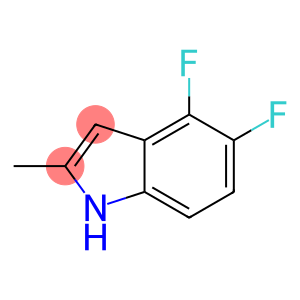
4,5-Difluoro-2-methylindole
4,5-Difluoro-2-methylindole
CAS: 85462-60-8
Molecular Formula: C9H7F2N
4,5-Difluoro-2-methylindole - Names and Identifiers
| Name | 4,5-Difluoro-2-methylindole |
| Synonyms | ,5-Difluoro-2-methylindole 4,5-DIFLUORO-2-METHYLINDOLE 4,5-Difluoro-2-methylindole 4,5-DIFLUORO-2-METHYL-1H-INDOLE 4,5-Difluoro-2-methyl-1H-indole 1H-Indole, 4,5-difluoro-2-methyl- |
| CAS | 85462-60-8 |
| InChI | InChI=1/C9H7F2N/c1-5-4-6-8(12-5)3-2-7(10)9(6)11/h2-4,12H,1H3 |
4,5-Difluoro-2-methylindole - Physico-chemical Properties
| Molecular Formula | C9H7F2N |
| Molar Mass | 167.16 |
| Density | 1.32g/cm3 |
| Boling Point | 275.4°C at 760 mmHg |
| Flash Point | 120.3°C |
| Vapor Presure | 0.00858mmHg at 25°C |
| Storage Condition | Sealed in dry,Room Temperature |
| Refractive Index | 1.6 |
4,5-Difluoro-2-methylindole - Risk and Safety
| Risk Codes | 36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. |
4,5-Difluoro-2-methylindole - Introduction
4,5-Difluoro-2-methylindole (4,5-Difluoro-2-methylindole) is an organic compound. It has a molecular formula of C9H7F2N and a molecular weight of 169.16g/mol.
4,5-Difluoro-2-methylindole is a white solid, practically insoluble in water at room temperature, but soluble in organic solvents such as ether, methylene chloride and benzene.
Its main use is as an intermediate in organic synthesis. It can be used to synthesize various compounds containing indole structure, such as drugs, pesticides and dyes. Indole structures have a wide range of applications in the field of medicine, because they can interact with proteins in vivo and play a therapeutic role.
The preparation method of 4,5-Difluoro-2-methylindole can be achieved by introducing a fluorine atom on the 2-methylindole molecule. One common method is to carry out the substitution reaction using a fluorinated reagent, such as hydrogen fluoride and a fluorite.
Regarding safety information, 4,5-Difluoro-2-methylindole has not been subjected to detailed toxicity studies. However, because it is an organic compound, it may have certain toxicity and potential danger. During handling and use, follow appropriate laboratory safety practices and use appropriate personal protective equipment, such as laboratory gloves and protective glasses.
4,5-Difluoro-2-methylindole is a white solid, practically insoluble in water at room temperature, but soluble in organic solvents such as ether, methylene chloride and benzene.
Its main use is as an intermediate in organic synthesis. It can be used to synthesize various compounds containing indole structure, such as drugs, pesticides and dyes. Indole structures have a wide range of applications in the field of medicine, because they can interact with proteins in vivo and play a therapeutic role.
The preparation method of 4,5-Difluoro-2-methylindole can be achieved by introducing a fluorine atom on the 2-methylindole molecule. One common method is to carry out the substitution reaction using a fluorinated reagent, such as hydrogen fluoride and a fluorite.
Regarding safety information, 4,5-Difluoro-2-methylindole has not been subjected to detailed toxicity studies. However, because it is an organic compound, it may have certain toxicity and potential danger. During handling and use, follow appropriate laboratory safety practices and use appropriate personal protective equipment, such as laboratory gloves and protective glasses.
Last Update:2024-04-09 21:04:16
Supplier List
Spot supply
Product Name: 4,5-Difluoro-2-methylindole Visit Supplier Webpage Request for quotationCAS: 85462-60-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 4,5-DIFLUORO-2-METHYLINDOLE Request for quotation
CAS: 85462-60-8
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 85462-60-8
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: 4,5-DIFLUORO-2-METHYLINDOLE Visit Supplier Webpage Request for quotation
CAS: 85462-60-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 85462-60-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 4,5-Difluoro-2-methylindole Visit Supplier Webpage Request for quotationCAS: 85462-60-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 4,5-DIFLUORO-2-METHYLINDOLE Request for quotation
CAS: 85462-60-8
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 85462-60-8
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: 4,5-DIFLUORO-2-METHYLINDOLE Visit Supplier Webpage Request for quotation
CAS: 85462-60-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 85462-60-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


