38260-54-7
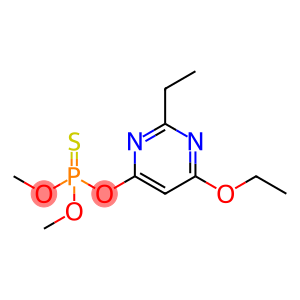
O-6-ethoxy-2-ethylpyrimidin-4-yl O,O-dimethylphosphorothioate
CAS: 38260-54-7
Molecular Formula: C10H17N2O4PS
38260-54-7 - Names and Identifiers
| Name | O-6-ethoxy-2-ethylpyrimidin-4-yl O,O-dimethylphosphorothioate |
| Synonyms | ekametg SATISFAR Etrimfos Ekamet G ENT 29126 ekametulv Ekamet ulv Etrimfos (ISO) Dimethyl-O-(2-ethyl-4-ethoxy-pyrimidinyl-6)-thionophosphate O-(6-ETHOXY-2-ETHYL-4-PYRIMIDINYL)-O,O-DIMETHYLTHIOPHOSPHATE O-6-ethoxy-2-ethylpyrimidin-4-yl O,O-dimethylphosphorothioate O-(6-ethoxy-2-ethylpyrimidin-4-yl) O,O-dimethyl phosphorothioate |
| CAS | 38260-54-7 |
| EINECS | 253-855-9 |
| InChI | InChI=1/C10H17N2O4PS/c1-5-8-11-9(15-6-2)7-10(12-8)16-17(18,13-3)14-4/h7H,5-6H2,1-4H3 |
38260-54-7 - Physico-chemical Properties
| Molecular Formula | C10H17N2O4PS |
| Molar Mass | 292.29 |
| Density | 1.195 g/cm3 (20℃) |
| Melting Point | -3.35℃ |
| Boling Point | approximate 77℃ |
| Flash Point | >100°C |
| Water Solubility | 40 mg l-1 (23-24 °C) |
| Solubility | Chloroform (Slightly), DMSO (Slightly), Methanol (Slightly) |
| Vapor Presure | 6.5 x 10-3 Pa (24 °C) |
| Appearance | liquid |
| Color | Colourless |
| Merck | 13,3923 |
| BRN | 7136389 |
| pKa | -0.67±0.32(Predicted) |
| Storage Condition | APPROX 4°C |
| Refractive Index | nD20 1.5068 |
| Physical and Chemical Properties | Flash point> 100°C storage conditions APPROX 4°C Merck 13,3923 pure colorless oil, M.P. -3.4 ℃, relative density of 1.195 (20 ℃), refractive index n20D1.5068, with acetone, chloroform, methanol, ethanol, hexanol, dimethyl sulfoxide, xylene miscible, solubility in water is 10g/L, 25 C hydrolysis half-life of 0.4d (pH = 3), 16d(pH = 6), 14d (pH = 9). Thiopyrimidine is not stable but is stable in dilute solutions in non-polar solvents, and suitable stabilizers are said to have been found. |
| Use | Efficient, broad-spectrum, non-systemic contact and stomach poison insecticide. It is mainly used for controlling Coleoptera, Lepidoptera, Hemiptera and cerclons on fruit trees, vegetables, paddy fields, potatoes, corn, olive and alfalfa. The effective application amount of 0.25~0.75/hm2, if the use of granules to control rice borer is 1~1.5/hm2, the effective period of 7 ~ 14d. |
38260-54-7 - Risk and Safety
| Risk Codes | R22 - Harmful if swallowed R50/53 - Very toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment. |
| Safety Description | S60 - This material and its container must be disposed of as hazardous waste. S61 - Avoid release to the environment. Refer to special instructions / safety data sheets. |
| UN IDs | UN3082 9/PG 3 |
| WGK Germany | 3 |
| RTECS | TF8350000 |
| Toxicity | LD50 in male rats, mice (mg/kg): 1800, 437 orally; LC50 in carp (mg/l): 13.6 (48 hr); 13.3 (96 hr) (Knutti) |
38260-54-7 - Reference Information
| EPA chemical information | Information provided by: ofmpub.epa.gov (external link) |
| toxicity | acute oral LD501800mg/kg in rats, acute percutaneous LD50 in rats> 2000 mg/kg, and rabbit> 500 mg/kg; The non-acting dose in subchronic test is 9 mg/kg. The LC50 of carp is 13.3mg/L (96h). |
| use | efficient, broad-spectrum, non-systemic contact and gastrotoxic insecticide. Mainly used in fruit trees, vegetables, rice fields, potatoes, corn, olives and alfalfa to control Coleoptera, Lepidoptera, Hemiptera, rodent pests. The effective application amount is 0.25~0.75kg/hm2, if the granule is used to control rice borer is 1~1.5kg/hm2, the effective period is 7~14 days. Efficient, broad-spectrum, non-systemic contact and stomach poison insecticide. Mainly used in fruit trees, vegetables, rice fields, potatoes, corn, olives and alfalfa to control Coleoptera, Lepidoptera, Hemiptera, rodent pests. The effective application amount is 0.25~0.75kg/hm2, if the granule is used to control rice borer is 1~1.5kg/hm2, the effective period is 7~14 days. |
| production method | preparation of propylamide hydrochloride 22g of propionitrile and 37g of ethanol are added to the reaction bottle, dried hydrogen chloride gas is introduced at 25~30 ℃ until 17.5g of hydrogen chloride gas is absorbed, ammonia gas is introduced at 10~15 ℃ after heat preservation reaction for 1h, and ammonia is stopped at pH 8~9, heat preservation reaction for 0.5h, cooling to below 0 ℃, filtering, and washing the filter residue with appropriate amount of ice ethanol. After the filtrate was concentrated, the propylene amine hydrochloride solid was obtained, which weighed 40g after drying, the content was 99%, and the yield was 91.1%,m.p. 129~130 ℃. Preparation of 2-Ethyl -4, 6-Dihydroxypyrimidine 33g of sodium hydroxide and 250mL of ethanol were added to the reaction flask, stirred, until the sodium hydroxide was completely dissolved, 30g of propamidine hydrochloric acid and 44g of diethyl malonate were added, the temperature was heated and refluxed for 2-3h, and the ethanol was recovered by vacuum distillation until it was evaporated. Add appropriate amount of water until the solid is completely dissolved, slowly add hydrochloric acid under cooling, acidify to pH 2~3, filter the precipitated solid, recrystallize with ammonia water and dry to obtain 38g of product with 98% content and 97.3% yield, m.p. 298~300 ℃ (decomposition). Preparation of 2-ethyl-4, 6-dichloropyrimidine 35g of 2-ethyl-4, 6-dihydroxypyrimidine, 100g of phosphorus pentachloride and 70mL of solvent were added to the reaction bottle, heated and refluxed for 3h, cooled and added ice water. Let it stand and separate the upper organic layer. After dissolving the organic layer, the organic layer was distilled under reduced pressure to obtain 37g of colorless liquid with 99.5% content and 85% yield. Preparation of 2-Ethyl -6-Chloro -4-Hydroxypyrimidine 35g of 2-ethyl -4,6-dichloropyrimidine, 30mL of concentrated hydrochloric acid, 40mL of water and 20mL of n-butanol were added to the reaction flask, heated and refluxed for 2h, cooled, precipitated, filtered, washed with water to be neutral, dried to obtain 28.5g of product with a content of more than 98.5%, with a yield of 90%, m.p. 162~163 ℃. Preparation of 2-ethyl-6-ethoxy-4-hydroxypyrimidine 25g of 2-ethyl-6-chloro-4-hydroxypyrimidine, 117g of sodium ethoxide (content 18%) and 0.5g of catalyst A are added to the reaction bottle, reacted at 70~75 ℃ for 3 hours, then ethanol is recovered, the residue is dissolved in water, acidified with concentrated hydrochloric acid, solid is precipitated, filtered, washed with water, and dried to obtain 24.5g of product, the content is 98%, m.p. 121~123 ℃. Synthesis of ethylsulfuron 20g of 2-ethyl -6-ethoxy -4-hydroxypyrimidine, 5.5g of sodium hydroxide and 45mL of solvent are added to the reaction bottle, heated and refluxed until anhydrous formation is generated, cooled to 45-50 ℃, 0.1g of catalyst B is added, 20g of O,O-dimethyl thiophosphoryl chloride is added dropwise, heat is kept dropwise and reacted at this temperature for 3 hours, water is added for washing, stratified, and the organic layer is dried with anhydrous sodium sulfate, filtration, filtrate added with appropriate amount of stabilizer and concentrated to obtain 35.5g of light yellow ethoxypyridoxine crude oil with 90.2% content and 93.8% yield. |
| toxic substance data | information provided by: pubchem.ncbi.nlm.nih.gov (external link) |
Last Update:2024-04-09 21:32:05
Supplier List
Multiple SpecificationsSpot supply
Product Name: Etrimfos solution Visit Supplier Webpage Request for quotationCAS: 38260-54-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: ETRIMFOS Request for quotation
CAS: 38260-54-7
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 38260-54-7
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Multiple Specifications
Product Name: Etrimfos Visit Supplier Webpage Request for quotationCAS: 38260-54-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Multiple SpecificationsSpot supply
Product Name: Etrimfos solution Visit Supplier Webpage Request for quotationCAS: 38260-54-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: ETRIMFOS Request for quotation
CAS: 38260-54-7
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 38260-54-7
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Multiple Specifications
Product Name: Etrimfos Visit Supplier Webpage Request for quotationCAS: 38260-54-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


