34552-83-5
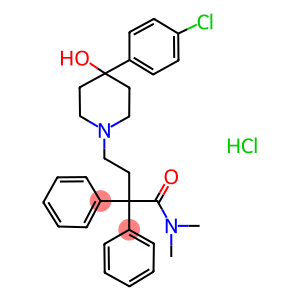
loperamide hydrochloride
CAS: 34552-83-5
Molecular Formula: C29H34Cl2N2O2
34552-83-5 - Names and Identifiers
34552-83-5 - Physico-chemical Properties
| Molecular Formula | C29H34Cl2N2O2 |
| Molar Mass | 513.5 |
| Density | 1.1905 (rough estimate) |
| Melting Point | 223-225°C |
| Boling Point | 647.2°C at 760 mmHg |
| Flash Point | 345.2°C |
| Solubility | Soluble in water (160 mg/ml), methanol (286 mg/ml), ethanol (53.7 mg/ml), DMSO (205 mg/ |
| Vapor Presure | 1.23E-17mmHg at 25°C |
| Appearance | neat |
| Color | White to Almost white |
| Merck | 14,5571 |
| pKa | 8.66(at 25℃) |
| Storage Condition | 2-8°C |
| Refractive Index | 1.6100 (estimate) |
| MDL | MFCD00058581 |
| Use | Used as antidiarrheal |
34552-83-5 - Risk and Safety
| Hazard Symbols | T - Toxic |
| Risk Codes | 25 - Toxic if swallowed |
| Safety Description | 45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) |
| UN IDs | UN 2811 6.1/PG 3 |
| WGK Germany | 3 |
| RTECS | TM4960000 |
| HS Code | 29333990 |
| Hazard Class | 6.1(b) |
| Packing Group | III |
| Toxicity | LD50 in mice (mg/kg): 75 s.c.; 28 i.p.; 105 orally; in rats (mg/kg): 185 orally (Niemegeers) |
34552-83-5 - Reference
| Reference Show more | 1. Li Mengwei, Yang Haiyang, Sun Wenyan, et al. Effects of Moringa oleifera leaves and its compound on defecation and gastrointestinal hormone levels in rats with constipation [J]. Chinese scientific and technological data catalogue-Chinese herbal medicine, 2019, 042(007):1291-1296. 2. [IF = 3.412] Yuefeng Zhang et al."Study on the Effective Material Basis and Mechanism of Traditional Chinese Medicine Prescription (QJC) Against Stress Diarrhea in Mice."Front Vet Sci. 2021; 8: 724491 3. [IF=4.932] Jing Xu et al."Atractyloside-A ameliorates spleen deficiency diarrhea by interfering with TLR4/MyD88/NF-κB signaling activation and regulating intestinal flora homeostasis."Int Immunopharmacol. 2022 Jun;107:108679 |
34552-83-5 - Nature
Open Data Verified Data
crystallized from isopropanol, melting point 222-223 °c.
34552-83-5 - Preparation Method
Open Data Verified Data
after the action of diphenylacetonitrile and sodium amino acid, ethylene oxide was introduced into the reaction. To the resulting product of the reaction, 36% hydrogen bromide in glacial acetic acid was added to give 4 Bromo -2,2-= phenyl butyric acid. Next, the reaction is carried out by oxidizing sulfoxide chloride, and then adding dimethylamine to toluene and water for acylation, and finally reacting with 4-monohydroxy-4-p-chlorophenylpiperidine to obtain loperamide.
34552-83-5 - Standard
Authoritative Data Verified Data
This product is N ,N-dimethyl-a,a-diphenyl -4-(p-chlorophenyl)-4-hydroyl-1-piperidinylbutanamide hydrochloride. The content of C29H33ClN202 • HC1 shall be between 98.0% and 102.0% calculated as dry.
34552-83-5 - Trait
Authoritative Data Verified Data
- This product is white or off-white crystalline powder; Almost odorless.
- This product is soluble in ethanol or glacial acetic acid, slightly soluble in water.
34552-83-5 - Use
Open Data Verified Data
long-acting anti-diarrhea drugs. Similar to morphine, can significantly inhibit intestinal peristalsis and diarrhea, but no morphine-like central inhibitory effect, also does not affect the intestinal cavity solute and water transport. Can be effective and safe control of acute and chronic diarrhea, can reduce ileostomy outflow and increase its hardness. It is suitable for acute Diarrhea and chronic Diarrhea caused by various causes, such as ulcerative colitis, nonspecific colitis, irritable bowel syndrome and the treatment of chronic Diarrhea caused by partial gastrectomy and hyperthyroidism.
34552-83-5 - Differential diagnosis
Authoritative Data Verified Data
- take this product, add methanol to make a solution containing 0401 mg per lml, according to UV-visible spectrophotometry (general) determination, at 265nm, 259nm and 253nm wavelength has the maximum absorption.
- The infrared absorption spectrum of this product should be consistent with that of the control (Spectrum set 1257).
34552-83-5 - Safety
Open Data Verified Data
loperamide hydrochloride: Mouse LDso(mg/kg): 75 subcutaneous injection, 105 by mouth. Rat oral LDso (mg/kg):185.
34552-83-5 - Differential diagnosis
Authoritative Data Verified Data
chlorine content
take about 15mg of this product, precision weighing, according to the oxygen flask combustion method (General 0703) for organic destruction, with lmol/L sodium hydroxide solution 20ml as the absorption liquid, once the combustion is completed, shake strongly for 15 minutes, rinse the bottle stopper and platinum wire with a small amount of water, add 1 drop of bromophenol blue indicator solution to the liquid, adjust with dilute nitric acid until the solution turns yellow, then add lml of dilute nitric acid, 20ml of ethanol and 5-10 drops of 1% diphenylhydrazine ethanol solution, titrate with mercury nitrate titration solution (0.005mol/L), with strong shaking near the end point, the solution was given a light rose-red color and the results of the titration were corrected with a blank test. Each 1 ml of mercury nitrate titration solution (0.005mol/L) is equivalent to 0.3545mg of C1. Chlorine content should be 13.52% ~ 14.20%.
Related substances
take this product O.lg, put in 100ml measuring flask, add methanol to dissolve and dilute to the scale, shake well, as a test solution; Take 1ml, put in 200ml measuring flask, dilute to the scale with methanol, as a control solution. Determined by high performance liquid chromatography (General 0512). Silica gel bonded with eighteen alkyl silane was used as the filler, and O.Olmol/L tetrabutylammonium hydrogen sulfate solution-acetonitrile-methanol (63:26:11) was used as the mobile phase. The detection wavelength was 220nm. The number of theoretical plates shall not be less than 3000 calculated by loperamide hydrochloride peak, and the separation degree between the main peak and the adjacent impurity peak shall meet the requirements. 20ul of control solution and 20ul of test solution were respectively injected into human liquid chromatograph, and the chromatogram was recorded to 2 times of the retention time of principal component peak. If there are impurity peaks in the chromatogram of the test solution, the area of a single impurity peak shall not be greater than 0.4 times (0.2%) of the area of the main peak of the control solution, the sum of each impurity peak area shall not be greater than the main peak area of the control solution (0.5%).
loss on drying
take this product, dry to constant weight at 105°C, weight loss shall not exceed 0.5% (General rule 0831).
ignition residue
This product l.Og, inspection according to law (General rule 0841), residual flooding shall not exceed 0.2%.
Heavy metals
The residue left under the item of taking the ignition residue shall not contain more than 20 parts per million of heavy metal when examined by law (General rule 0821, Law II).
34552-83-5 - Content determination
Authoritative Data Verified Data
take about 0.4g of this product, weigh it accurately, add 50ml of ethanol and 5.0ml of O.Olmol/L hydrochloric acid solution, shake to dissolve. Potentiometric titration (General 0701), with sodium hydroxide titration solution (0.lmol/L) titration, record the volume difference of two breakthrough point consumption titration solution, each lml sodium hydroxide titration solution (0.1 mol/L) corresponds to 51.35mg of C29H33C1N202. HCl.
34552-83-5 - Category
Authoritative Data Verified Data
antidiarrheal drugs.
34552-83-5 - Storage
Authoritative Data Verified Data
light shielding, sealed storage.
34552-83-5 - Loperamide Hydrochloride Capsules
Authoritative Data Verified Data
This product contains loperamide hydrochloride (C29H33C1N202 • HCl) should be 90.0% ~ 110.0% of label amount.
trait
The content of this product is white or white powder.
identification
In the chromatogram recorded under the content determination item, the retention time of the main peak of the test solution should be consistent with the retention time of the main peak of the control solution.
examination
- Content uniformity: Take 1 capsule of this product, place the content and capsule shell in the same 50ml plug Erlenmeyer flask, add 20ml methanol accurately, shake for 30 minutes, and centrifuge, take the supernatant as the test solution, according to the method under the content determination, should comply with the provisions (General 0941).
- dissolution of this product, according to the dissolution and release determination method (General rule 0931 The first method), with PH 4.7 acetate buffer solution (1 mol/L acetic acid solution 200ml, add water 600ml, mix well, adjust pH value to 4.70±0.05 with lmol/L sodium hydroxide solution, dilute to 100ml with water) 500ml as dissolution medium, rotate at RPM, operate according to law, take appropriate amount of solution after 30 minutes, filter, take the filtrate as the test solution; Take the loperamide hydrochloride reference about 20mg, precision weighing, put in 100ml measuring flask, add appropriate amount of methanol to dissolve, dilute to scale with dissolution medium, shake; Take 2ml with precision, put in 100ml measuring flask, dilute to scale with dissolution medium, shake well, as a reference solution. Take the above two kinds of solution, according to the method under the content determination item, calculate the dissolution amount of each grain. The limit is 80% of the labeled amount and shall be in accordance with the provisions.
- others should comply with the relevant provisions under the capsule (General 0103).
Content determination
- measured by high performance liquid chromatography (General 0512).
- chromatographic conditions and system suitability test using eighteen alkyl silane bonded silica as filler; 0.Olmol/L tetrabutylammonium bisulfate solution-acetonitrile-methanol (63:26:11) as mobile phase; The detection wavelength was 220mn. The number of theoretical plates is not less than 3000 calculated from the loperamide hydrochloride peak.
- determination Method: Take 5 capsules of this product, place the contents and the capsule shell in the same plug Erlenmeyer flask, add 100ml methanol accurately, shake for 30 minutes, centrifuge for 5 minutes, and rotate at 3500 rpm, the supernatant was taken as the test solution, and 20ul was injected into the liquid chromatograph for precise measurement. The chromatogram was recorded, methanol was added to dissolve and quantitatively diluted to contain O per 1 ml. lmg solution, the same method for determination. According to the external standard method to calculate the peak area, that is.
category
with loperamide hydrochloride.
specification
2mg
storage
sealed and stored in a dry place.
Supplier List
CAS: 34552-83-5
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
CAS: 34552-83-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 34552-83-5
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 34552-83-5
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 34552-83-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 34552-83-5
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
CAS: 34552-83-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 34552-83-5
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 34552-83-5
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 34552-83-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025




