319-78-8
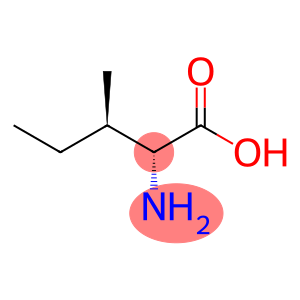
D-Isoleucine
CAS: 319-78-8
Molecular Formula: C6H13NO2
319-78-8 - Names and Identifiers
| Name | D-Isoleucine |
| Synonyms | D-Ile D-Ile-OH H-D-Ile-OH H-D-ILE-OH D-Isoleucine D-ISOLEUCINE D(-)-ISOLEUCINE [-]-2-AMINO-3-METHYLPENTANOIC ACID (2R,3R)-2-Amino-3-methylvaleric acid (2R,3R)-2-Amino-3-methylpentanoic acid |
| CAS | 319-78-8 |
| EINECS | 206-269-2 |
| InChI | InChI=1/C6H13NO2/c1-3-4(2)5(7)6(8)9/h4-5H,3,7H2,1-2H3,(H,8,9) |
319-78-8 - Physico-chemical Properties
| Molecular Formula | C6H13NO2 |
| Molar Mass | 131.17 |
| Density | 1.1720 (estimate) |
| Melting Point | 272-274 °C |
| Boling Point | 225.8±23.0 °C(Predicted) |
| Specific Rotation(α) | -37 º (c=5, 1 N HCl 24 ºC) |
| Solubility | Solubility in water: 41 · 2g/L (25°C) |
| Appearance | Solid |
| Color | White to Off-White |
| pKa | 2.57±0.24(Predicted) |
| Storage Condition | Sealed in dry,Room Temperature |
| Refractive Index | -40.5 ° (C=4, 6mol/L |
| MDL | MFCD00064221 |
| Physical and Chemical Properties | White crystals or crystalline powder, odorless, slightly bitter taste; Soluble in water, ethanol-soluble, insoluble in ether; Decomposition point of 283-284 ℃; specific rotation [α]20D-12.4 °(0.5-2.0 mg/ml,H2O),[α]25D-30.5 °(0.5-2.0 mg/ml,5 mol/L HCl). |
319-78-8 - Risk and Safety
| Hazard Symbols | Xn - Harmful |
| Risk Codes | 40 - Limited evidence of a carcinogenic effect |
| Safety Description | S24/25 - Avoid contact with skin and eyes. S36 - Wear suitable protective clothing. S22 - Do not breathe dust. |
| WGK Germany | 3 |
| RTECS | NR4700000 |
| HS Code | 29224995 |
319-78-8 - Reference
| Reference Show more | 1. [IF=3.361] Xiao Lin et al."Applying molecular networking for targeted isolation of depsipeptides."Rsc Adv. 2021 Jan;11(5):2774-2782 |
319-78-8 - Standard
Authoritative Data Verified Data
This product is L-2-amino -3-methyl pentanoic acid. The content of C6H13N02F was less than 98.5% based on the dry product.
319-78-8 - Trait
Authoritative Data Verified Data
- This product is white crystal or crystalline powder; Odorless.
- This product is slightly soluble in water, and almost insoluble in ethanol or ether.
specific rotation
take this product, precision weighing, add 6mol/L hydrochloric acid solution to dissolve and quantitatively dilute to make a solution containing about 40mg per lml, and determine according to law (General 0621), the specific rotation was 38.9 ° to 41.8 °.
319-78-8 - Differential diagnosis
Authoritative Data Verified Data
- appropriate amounts of this product and the isoleucine reference substance are added respectively to water for dissolution and dilution to prepare a solution containing about 0.4mg per 1 ml as the test solution and the reference solution. According to the chromatographic condition test under other amino acids, the position and color of the main spot of the test solution should be the same as that of the reference solution.
- The infrared absorption spectrum of this product should be consistent with that of the control (Spectrum set 894).
319-78-8 - Exam
Authoritative Data Verified Data
acidity
take 0.20g of this product, add 20ml of water to dissolve, and then measure it according to law (General rule 0631). The pH value should be 5.5~6.5.
transmittance of solution
take 0.5g of this product, add water 20ml to dissolve, according to UV-visible spectrophotometry (General rule 0401), measure the transmittance at 430nm wavelength, not less than 98.0%.
chloride
take 0.25g of this product and check it according to law (General rule 0801). Compared with the control solution made of 0.02% of standard sodium chloride solution, it should not be more concentrated ().
sulfate
take l.Og of this product and check it according to law (General rule 0802). Compared with the control solution made of 0.02% of standard potassium sulfate solution, it should not be more concentrated ().
ammonium salt
take 0.10g of this product and check it according to law (General rule 0808). Compared with the control solution made of 2.0 ml of standard gasification ammonium solution, it shall not be deeper (0.02%).
other amino acids
take an appropriate amount of this product, add water to dissolve and dilute to make a solution containing about 20mg per lml as a test solution; Take 1ml for precision measurement and put it in a 200ml measuring flask, dilute to the scale with water, shake well, as a control solution; Take the appropriate amount of isoleucine reference and valine reference respectively, and put them in the same measuring flask, water was added to dissolve and diluted to prepare a solution containing about 0.4mg each per 1ml as a system-suitable solution. According to the thin layer chromatography (General 0502) test, absorb the above three solutions each 5 u1, respectively, on the same silica gel G thin layer plate, with n-butanol-water-glacial acetic acid (3:1:1) to develop, spread, dry, spray with ninhydrin in acetone solution (1-50), heat at 80°C until spots appear, and immediately inspect. The control solution should show a clear spot, and the system applicable solution should show two completely separated spots. If the test solution shows impurity spots, the color should not be deeper (0.5%) than the main spot of the control solution.
loss on drying
take this product, dry at 105°C for 3 hours, loss of weight shall not exceed 0.2% (General rule 0831).
ignition residue
take l.Og of this product and check it according to law (General rule 0841). The residue left shall not exceed 0.1%.
Iron Salt
take 2.0g of this product and check it according to law (General rule 0807). Compared with the control solution made of 0.001% of standard iron solution, it should not be deeper ().
Heavy metals
The residue left under the item of taking the ignition residue shall not contain more than 10 parts per million of heavy metal when examined by law (General Principles 0821, Law II).
arsenic salt
take 2.0g of this product, add 5ml of water, add 1ml of sulfuric acid and 10ml of sulfurous acid, heat to a volume of about 2ml in a water bath, add 5ml of water, ammonia Test Solution was added dropwise until the indicated solution of Phenolphthalein was neutral. Hydrochloric acid (0822) was added and water was added to make it 0.0001%.
bacterial endotoxin
take this product and check it according to law (General rule 1143). The amount of endotoxin in each lg isoleucine should be less than 20EU. (For injection)
319-78-8 - Content determination
Authoritative Data Verified Data
take this product about 0.10g, precision weighing, add anhydrous formic acid 1 ml dissolved, add glacial acetic acid 25ml, according to the potential titration method (General rule 0701), with perchloric acid titration solution (0.1 mol/L) titration, and the results of the titration were corrected with a blank test. Each 1 ml of perchloric acid titration solution (0.1 mol/L) corresponds to 13.12mg of C6H13Na2.
319-78-8 - Category
Authoritative Data Verified Data
amino acid drugs.
319-78-8 - Storage
Authoritative Data Verified Data
light shielding, sealed storage.
319-78-8 - Reference Information
| EPA chemical information | Information provided by: ofmpub.epa.gov (external link) |
| Overview | D-isoleucine is an important organic chiral source, which is increasingly widely used in the field of medicine. As an optically active organic acid, It has an irreplaceable role in the asymmetric synthesis of some chiral compounds. It is mainly used in the preparation of chiral drugs, chiral additives, chiral additives, etc, at the same time, it is also an important intermediate and pharmaceutical supplement for the synthesis of antibacterial, anti-tumor and new broad-spectrum antibiotics. |
| biological activity | D-Isoleucine ((R)-Isoleucine) is a stereoisomer of isoleucine, which is a selective activator of Asc-1 antitransport protein and can enhance the long-term synergy of hippocampal CA1-CA3 by releasing endogenous D-serine. |
| use | used for biochemical research and nutritional supplements in medicine. |
| Production method | Using L-isoleucine as raw material, after acylation with acetic anhydride, the product is treated with acylase to separate L-isoleucine and then hydrolyzed. |
Supplier List
CAS: 319-78-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 319-78-8
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 319-78-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 319-78-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 319-78-8
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 319-78-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025


