313546-16-6
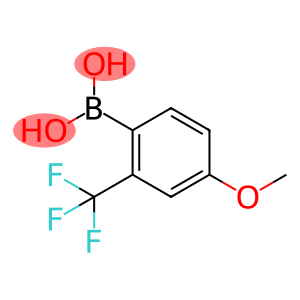
4-Methoxy-2-(trifluoromethyl)benzeneboronic acid
CAS: 313546-16-6
Molecular Formula: C8H8BF3O3
313546-16-6 - Names and Identifiers
313546-16-6 - Physico-chemical Properties
| Molecular Formula | C8H8BF3O3 |
| Molar Mass | 219.95 |
| Density | 1.36±0.1 g/cm3(Predicted) |
| Melting Point | 168-172 °C |
| Boling Point | 314.1±52.0 °C(Predicted) |
| Flash Point | 143.775°C |
| Vapor Presure | 0mmHg at 25°C |
| Appearance | powder to crystal |
| Color | White to Almost white |
| pKa | 8.54±0.58(Predicted) |
| Storage Condition | 2-8°C |
| Refractive Index | 1.465 |
| MDL | MFCD03095346 |
313546-16-6 - Risk and Safety
| WGK Germany | 3 |
313546-16-6 - Reference Information
| application | 4-methoxy-2-(trifluoromethyl) phenylboronic acid can be used as an intermediate in organic synthesis and a pharmaceutical intermediate for laboratory research and development and chemical production. |
| preparation | 3.0g (8.85mmol) potassium phosphate heptahydrate, 1.50g (5.90mmol) pinacol borate B2(pin)2, 12mg (0.015mmol) Xphos-Pd-G2 and 4mg (0.008mmol) Xphos were sequentially added to the reaction bottle, and 6mL of ethanol was added to stir evenly, add 0.36mL (2.95mmol) 4-methoxy-2-(trifluoromethyl) chlorobenzene and react at room temperature for 1 hour. 5mL ethyl acetate was added to the reaction solution to dilute, filtered by diatomite, washed by ethyl acetate, combined filtrate, concentrated under reduced pressure to obtain crude product, separated by silica gel column chromatography, and eluted by petroleum ether-ethyl acetate to obtain 4-methoxy-2-(trifluoromethyl) phenylboronic acid pinacol ester. 4-methoxy-2-(trifluoromethyl) phenylboronic acid pinacol ester was added to the reaction bottle, and dilute HCl was added dropwise for hydrolysis. The solution first produces precipitation, and gradually disappears with the precipitation, and the pH value of the system is adjusted to 1. Drop the NaOH solution with a mass fraction of 25% to the solution to a pH value of 13, and stir for 1 h. The organic phase is extracted with 15 mL of NaOH with a mass fraction of 10%, the aqueous phase is combined, and the lye is extracted twice with 15mL of THF. Adjust the pH value of the obtained lye with dilute HCl, turbidity begins to occur, flocs slowly appear, and adjust the pH value to 5.0. The aqueous phase was extracted with 70 mLTHF, and the organic phase was spin-dried and purified to obtain 4-methoxy-2-(trifluoromethyl) phenylboronic acid. |
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: 4-Methoxy-2-(trifluoromethyl)phenylboronic acid Visit Supplier Webpage Request for quotationCAS: 313546-16-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 4-METHOXY-2-(TRIFLUOROMETHYL)PHENYLBORONIC ACID Request for quotation
CAS: 313546-16-6
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 313546-16-6
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: 4-Methoxy-2-(trifluoromethyl)phenylboronic acid Visit Supplier Webpage Request for quotation
CAS: 313546-16-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 313546-16-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 4-Methoxy-2-(trifluoromethyl)phenylboronic acid Visit Supplier Webpage Request for quotationCAS: 313546-16-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 4-METHOXY-2-(TRIFLUOROMETHYL)PHENYLBORONIC ACID Request for quotation
CAS: 313546-16-6
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 313546-16-6
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: 4-Methoxy-2-(trifluoromethyl)phenylboronic acid Visit Supplier Webpage Request for quotation
CAS: 313546-16-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 313546-16-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


