31277-98-2
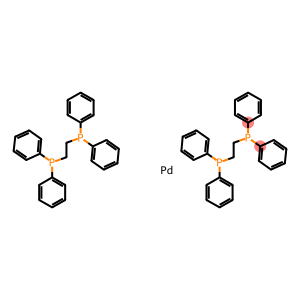
Bis[1,2-bis(diphenylphosphino)ethane]-palladium
CAS: 31277-98-2
Molecular Formula: C52H48P4Pd
31277-98-2 - Names and Identifiers
| Name | Bis[1,2-bis(diphenylphosphino)ethane]-palladium |
| Synonyms | Pd(dppe)2 PD(DIPHOS)2 BIS(DIPHOS)PALLADIUM(0) Bisbisdiphenylphosphinoethanepalladium bis(ethylenebis(diphenylphosphine))palladium BIS[1,2-BIS(DIPHENYLPHOSPHINO)ETHANE]-PALLADIUM Bis[1,2-bis(diphenylphosphino)ethane]-palladium Bis[1,2-bis(diphenylphosphino)ethane]palladium(0) BIS[1,2-BIS(DIPHENYLPHOSPHINO)ETHANE]PALLADIUM(0) Bis[1,2-bis(diphenylphosphino)ethane]palladium (0) |
| CAS | 31277-98-2 |
| EINECS | 625-554-6 |
| InChI | InChI=1/2C26H24P2.Pd/c2*1-5-13-23(14-6-1)27(24-15-7-2-8-16-24)21-22-28(25-17-9-3-10-18-25)26-19-11-4-12-20-26;/h2*1-20H,21-22H2 |
| InChIKey | UTBLWXFSGOYWOH-UHFFFAOYSA-R |
31277-98-2 - Physico-chemical Properties
| Molecular Formula | C52H48P4Pd |
| Molar Mass | 903.25 |
| Melting Point | 219-225 °C |
| Boling Point | 514.8°C at 760 mmHg |
| Flash Point | 281.7°C |
| Water Solubility | insoluble |
| Vapor Presure | 3.38E-10mmHg at 25°C |
| Appearance | Yellow to or orange crystals |
| Color | orange |
| Storage Condition | -20°C |
| Sensitive | Air Sensitive |
| MDL | MFCD00009880 |
31277-98-2 - Risk and Safety
| Risk Codes | R20/21/22 - Harmful by inhalation, in contact with skin and if swallowed. R36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S36 - Wear suitable protective clothing. S37/39 - Wear suitable gloves and eye/face protection S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. |
| UN IDs | 1479 |
| WGK Germany | 3 |
| HS Code | 28439000 |
| Hazard Class | 5.1 |
| Packing Group | III |
31277-98-2 - Reference Information
| Application | Bis (1, 2-bis (diphenylphosphine) ethane) palladium is mainly used as a catalyst in organic synthesis reactions. It can be prepared by heating 1, 2-bis (diphenylphosphine) ethane and palladium dichloride in DMSO. It has been reported in the literature that bis (1, 2-bis (diphenylphosphine) ethane) palladium is used to prepare fullerene pyrrole metal complexes (PyrC60)-Pd(dppe). |
| use | bis (1,2-bis (diphenylphosphine) ethane) palladium is a precious metal organic substance and can be used as a catalyst containing palladium. |
| preparation | under the protection of nitrogen, 0.2 gPdCl2,1.12gPh2PCH2CH2PPh2 and 12ml dimethyl sulfoxide are mixed and heated to 140 ℃ to make them into a uniform solution. at this time, the solution is light red, the oil bath is removed, stirred intensely for 30min, then 0.3ml(0.226g) hydrazine hydrate is slowly added dropwise, the reaction is intense, nitrogen is generated, and the solution turns dark brown, cooling in a water bath, precipitating brown crystals at about 125°C, slowly cooling to room temperature, centrifuging, washing twice with 5ml of ethanol and 5ml of ether, and vacuum drying to obtain orange crystals with a yield of about 78.2%. |
Last Update:2024-04-09 20:52:54
Supplier List
Multiple SpecificationsSpot supply
Product Name: Bis[1,2-bis(diphenylphosphino)ethane]palladium(0) Visit Supplier Webpage Request for quotationCAS: 31277-98-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Bis[1,2-bis(diphenylphosphino)ethane]palladium(0) Request for quotation
CAS: 31277-98-2
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 31277-98-2
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Spot supply
Product Name: Bis[1,2-bis(diphenylphosphino)ethane]palladium Visit Supplier Webpage Request for quotationCAS: 31277-98-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Multiple SpecificationsSpot supply
Product Name: Bis[1,2-bis(diphenylphosphino)ethane]palladium(0) Visit Supplier Webpage Request for quotationCAS: 31277-98-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Bis[1,2-bis(diphenylphosphino)ethane]palladium(0) Request for quotation
CAS: 31277-98-2
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 31277-98-2
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Spot supply
Product Name: Bis[1,2-bis(diphenylphosphino)ethane]palladium Visit Supplier Webpage Request for quotationCAS: 31277-98-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


