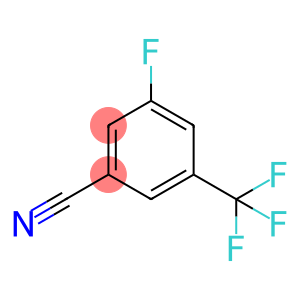
3-fluoro-5-(trifluoromethyl)benzonitrile
3-fluoro-5-(trifluoromethyl)benzonitrile
CAS: 149793-69-1
Molecular Formula: C8H3F4N
3-fluoro-5-(trifluoromethyl)benzonitrile - Names and Identifiers
| Name | 3-fluoro-5-(trifluoromethyl)benzonitrile |
| Synonyms | 3-Cyano-5-fluorobenzotrifluoride 3-CYANO-5-FLUOROBENZOTRIFLUORIDE à,à,à,5-tetrafluoro-m-tolunitrile 3-FLUORO-5-(TRIFLUOROMETHYL)BENZONITRILE 3-fluoro-5-(trifluoromethyl)benzonitrile alpha,alpha,alpha,5-Tetrafluoro-m-tolunitrile 5-Fluoro-3-(trifluoroMethyl)benzenecarbonitrile 3-Cyano-5-fluorobenzotrifluoride~alpha,alpha,alpha,5-Tetrafluoro-m-tolunitrile |
| CAS | 149793-69-1 |
| InChI | InChI=1/C8H3F4N/c9-7-2-5(4-13)1-6(3-7)8(10,11)12/h1-3H |
| InChIKey | AYNUKEDZAYOEGG-UHFFFAOYSA-N |
3-fluoro-5-(trifluoromethyl)benzonitrile - Physico-chemical Properties
| Molecular Formula | C8H3F4N |
| Molar Mass | 189.11 |
| Density | 1.35 |
| Boling Point | 179.9±35.0 °C(Predicted) |
| Flash Point | 65°(149°F) |
| Solubility | Chloroform (Sparingly), Ethyl Acetate (Slightly) |
| Vapor Presure | 0.919mmHg at 25°C |
| Appearance | Oil |
| Color | Clear Colourless |
| BRN | 7703957 |
| Storage Condition | 2-8°C |
| Refractive Index | 1.446 |
3-fluoro-5-(trifluoromethyl)benzonitrile - Risk and Safety
| Risk Codes | R20/21/22 - Harmful by inhalation, in contact with skin and if swallowed. R36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) S36 - Wear suitable protective clothing. |
| UN IDs | 3276 |
| Hazard Note | Toxic |
| Hazard Class | 6.1 |
| Packing Group | III |
3-fluoro-5-(trifluoromethyl)benzonitrile - Reference Information
| description | 3-fluoro-5-(trifluoromethyl) benzonitrile is a white to pale yellow crystalline powder. Halogenated Benzonitrile is an important organic intermediate, which is mainly used for the preparation of medicines and pesticides. It is widely used in the manufacturing process of dyes, engineering plastics and photosensitive materials. Benzonitrile is an important organic intermediate, which is mainly used for the preparation of medicines and pesticides. It is widely used in the manufacturing process of dyes, engineering plastics and photosensitive materials. |
| preparation | The traditional synthesis methods of halogenated benzonitrile include light halogenation, ammonia oxidation, phase transfer catalytic synthesis or reaction of carboxylic acid with urea. In this paper, substituted benzaldehyde as the starting material, then into 3-fluoro-5-(trifluoromethyl) benzaldehyde oxime, and then the oxime and hydroxylamine reaction dehydration substituted benzonitrile, although the route more steps, but the yield of each step is high, and the product is easy to separate and purify, which is a feasible route [1]. The reaction formula for the synthesis of 3-fluoro-5-(trifluoromethyl) benzonitrile is shown below: Fig. 1 The Reaction formula for the synthesis of 3-fluoro-5-(trifluoromethyl) benzonitrile, 3-fluorine -5-(trifluoromethyl) benzaldehyde and hydroxylamine hydrochloride with a certain ratio of the amount of substances were added to the condenser tube and the electric stirred three-necked flask, and the reaction was maintained at 70~75 ℃ for 30min, then, 20 (mass fraction) aqueous sodium carbonate solution was slowly added dropwise and stirred, and bubbles were generated (which can be checked with lime water). Until the reaction slows down, the pH is adjusted to 8-9, and the dropwise addition of the aqueous sodium carbonate solution is stopped. With continued stirring, a light red precipitate appeared, which was allowed to stand until the light red color gradually turned milky white. The precipitate was filtered off, washed with water for 2-3 times, and dried to obtain a milky white solid powder 3-fluoro-5-(trifluoromethyl) benzaldehyde oxime, which was weighed for use. Add 3-fluoro-5-(trifluoromethyl) benzaldehyde oxime and acetic anhydride into a three-necked bottle according to the ratio of a certain amount of substances, heat to 110~120 ℃, react for 3H, the reaction solution was light orange liquid, which was cooled and allowed to stand. White crystals appeared. The crystals were obtained by Suction filtration and dissolved in alcohol. The insoluble matters were removed by hot filtration and the filtrate was separated by extraction with water to obtain clean 3-fluoro-5-(trifluoromethyl) benzonitrile. |
Last Update:2024-04-09 15:16:45
Supplier List
Spot supply
Product Name: 3-Fluoro-5-(trifluoromethyl)benzonitrile Visit Supplier Webpage Request for quotationCAS: 149793-69-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 3-FLUORO-5-(TRIFLUOROMETHYL)BENZONITRILE Request for quotation
CAS: 149793-69-1
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 149793-69-1
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Spot supply
Product Name: 3-Fluoro-5-(trifluoromethyl)benzonitrile Visit Supplier Webpage Request for quotationCAS: 149793-69-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 3-FLUORO-5-(TRIFLUOROMETHYL)BENZONITRILE Request for quotation
CAS: 149793-69-1
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 149793-69-1
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
View History

