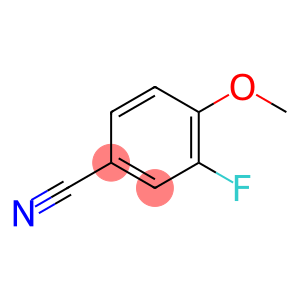
3-fluoro-4-methoxybenzonitrile
3-fluoro-4-methoxybenzonitrile
CAS: 331-62-4
Molecular Formula: C8H6FNO
3-fluoro-4-methoxybenzonitrile - Names and Identifiers
| Name | 3-fluoro-4-methoxybenzonitrile |
| Synonyms | 4-Cyano-2-fluoroanisole Fluoromethoxybenzonitrile Fluoromethoxybenzonitrile5 3-FLUORO-4-METHOXYBENZONITRILE 3-fluoro-4-methoxybenzonitrile 3-fluoro-4-methoxy-carbonitrile |
| CAS | 331-62-4 |
| InChI | InChI=1/C8H6FNO/c1-11-8-3-2-6(5-10)4-7(8)9/h2-4H,1H3 |
| InChIKey | FEEOVAOEPGQDTJ-UHFFFAOYSA-N |
3-fluoro-4-methoxybenzonitrile - Physico-chemical Properties
| Molecular Formula | C8H6FNO |
| Molar Mass | 151.14 |
| Density | 1.18±0.1 g/cm3(Predicted) |
| Melting Point | 97-101 °C |
| Boling Point | 96-98°C/1mm |
| Flash Point | 96-98°C/1mm |
| Vapor Presure | 0.0173mmHg at 25°C |
| Appearance | Crystallization |
| Color | White to Almost white |
| Storage Condition | Sealed in dry,Room Temperature |
| Refractive Index | 1.505 |
| MDL | MFCD00215835 |
3-fluoro-4-methoxybenzonitrile - Risk and Safety
| Risk Codes | R20/21/22 - Harmful by inhalation, in contact with skin and if swallowed. R36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S23 - Do not breathe vapour. S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. S36 - Wear suitable protective clothing. S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. |
| UN IDs | 3439 |
| WGK Germany | 3 |
| HS Code | 29269090 |
| Hazard Note | Toxic |
| Hazard Class | 6.1 |
| Packing Group | III |
3-fluoro-4-methoxybenzonitrile - Introduction
It is an organic compound with the chemical formula C8H6FNO. The following is a description of its nature, use, preparation and safety information:
Nature:
-Appearance: Colorless or light yellow crystalline solid.
-Melting point: about 74-75 degrees Celsius.
-Boiling point: About 242 degrees Celsius.
-Solubility: Soluble in some organic solvents, such as ethanol, methanol and dichloromethane.
Use:
-is useful as an intermediate and starting material in organic synthesis.
-It can be used to prepare compounds such as anticancer drugs, pesticides and pharmaceutical intermediates.
Method:
-or is prepared generally by introducing a fluorine atom into 4-methoxybenzonitrile. This can be achieved by reacting fluorinating reagents such as brominated and iodinated fluorides, or by reacting antimony pentafluoride with 4-methoxybenzonitrile in the presence of hydrofluoric acid.
Safety Information:
-It has limited data on toxicity and danger, so be careful when using it.
-As a chemical, it may be irritating to the eyes, skin and respiratory system.
-Observe proper laboratory procedures during use or handling, and wear appropriate personal protective equipment, such as gloves, protective glasses and laboratory coats.
-If you accidentally inhale or contact, you should immediately leave the scene, clean the affected area with plenty of water, and seek medical attention in time.
Nature:
-Appearance: Colorless or light yellow crystalline solid.
-Melting point: about 74-75 degrees Celsius.
-Boiling point: About 242 degrees Celsius.
-Solubility: Soluble in some organic solvents, such as ethanol, methanol and dichloromethane.
Use:
-is useful as an intermediate and starting material in organic synthesis.
-It can be used to prepare compounds such as anticancer drugs, pesticides and pharmaceutical intermediates.
Method:
-or is prepared generally by introducing a fluorine atom into 4-methoxybenzonitrile. This can be achieved by reacting fluorinating reagents such as brominated and iodinated fluorides, or by reacting antimony pentafluoride with 4-methoxybenzonitrile in the presence of hydrofluoric acid.
Safety Information:
-It has limited data on toxicity and danger, so be careful when using it.
-As a chemical, it may be irritating to the eyes, skin and respiratory system.
-Observe proper laboratory procedures during use or handling, and wear appropriate personal protective equipment, such as gloves, protective glasses and laboratory coats.
-If you accidentally inhale or contact, you should immediately leave the scene, clean the affected area with plenty of water, and seek medical attention in time.
Last Update:2024-04-09 20:49:11
Supplier List
Spot supply
Product Name: 3-Fluoro-4-methoxybenzonitrile Visit Supplier Webpage Request for quotationCAS: 331-62-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 3-Fluoro-4-methoxybenzonitrile Request for quotation
CAS: 331-62-4
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 331-62-4
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: 3-Fluoro-4-Methoxybenzonitrile Visit Supplier Webpage Request for quotation
CAS: 331-62-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 331-62-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 3-Fluoro-4-methoxybenzonitrile Visit Supplier Webpage Request for quotationCAS: 331-62-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 3-Fluoro-4-methoxybenzonitrile Request for quotation
CAS: 331-62-4
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 331-62-4
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: 3-Fluoro-4-Methoxybenzonitrile Visit Supplier Webpage Request for quotation
CAS: 331-62-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 331-62-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


