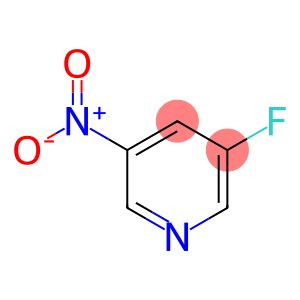
3-Fluoro-5-nitropyridine
3-Fluoro-5-nitropyridine
CAS: 1060804-39-8
Molecular Formula: C5H3FN2O2
3-Fluoro-5-nitropyridine - Names and Identifiers
| Name | 3-Fluoro-5-nitropyridine |
| Synonyms | 3-Fluoro-5-nitropyridine Pyridine, 3-fluoro-5-nitro- |
| CAS | 1060804-39-8 |
| InChI | InChI=1S/C5H3FN2O2/c6-4-1-5(8(9)10)3-7-2-4/h1-3H |
3-Fluoro-5-nitropyridine - Physico-chemical Properties
| Molecular Formula | C5H3FN2O2 |
| Molar Mass | 142.09 |
| Density | 1.439±0.06 g/cm3(Predicted) |
| Boling Point | 203.9±20.0 °C(Predicted) |
| pKa | -1.23±0.20(Predicted) |
| Storage Condition | Inert atmosphere,Room Temperature |
3-Fluoro-5-nitropyridine - Introduction
3-fluoro-5-nitropyridine is an organic compound with the chemical formula C5H2FN3O2. The following is an introduction to some of its properties, uses, methods and safety information:
Nature:
-Appearance: 3-fluoro-5-nitropyridine is a colorless to pale yellow solid.
-Melting point: Its melting point range is 45-48 degrees Celsius.
-Stability: 3-fluoro-5-nitropyridine is stable at room temperature and is not easily affected by moisture.
-Solubility: It has good solubility in some organic solvents, such as ethanol, dimethyl sulfoxide, etc.
Use:
-Military use: 3-fluoro-5-nitropyridine can be used to prepare explosives containing nitrogen and oxygen, with high combustion speed and explosive power.
-Chemical reagent: It can also be used as a chemical reagent in organic synthesis to synthesize other compounds.
Preparation Method:
There are two main methods for the preparation of 3-fluoro-5-nitropyridine:
-Through nitrification reaction: pyridine is reacted with concentrated nitric acid, and the reaction product is fluorinated with hydrogen fluoride gas to obtain 3-fluoro-5-nitropyridine.
-Through nitric acid esterification reaction: reacting pyridine with nitrate ester, and then fluorinating the reaction product with hydrogen fluoride gas, 3-fluoro-5-nitropyridine can be prepared.
Safety Information:
-3-fluoro-5-nitropyridine is an organic compound, which has high toxicity and explosiveness and should be used with care.
-Avoid contact with skin, eyes and respiratory tract, and wear protective equipment.
-Keep away from fire and heat sources during use and storage, and avoid severe impact and friction.
Please note that when conducting chemical experiments or preparing compounds, correct operating procedures should be followed and laboratory safety standards should be strictly observed.
Nature:
-Appearance: 3-fluoro-5-nitropyridine is a colorless to pale yellow solid.
-Melting point: Its melting point range is 45-48 degrees Celsius.
-Stability: 3-fluoro-5-nitropyridine is stable at room temperature and is not easily affected by moisture.
-Solubility: It has good solubility in some organic solvents, such as ethanol, dimethyl sulfoxide, etc.
Use:
-Military use: 3-fluoro-5-nitropyridine can be used to prepare explosives containing nitrogen and oxygen, with high combustion speed and explosive power.
-Chemical reagent: It can also be used as a chemical reagent in organic synthesis to synthesize other compounds.
Preparation Method:
There are two main methods for the preparation of 3-fluoro-5-nitropyridine:
-Through nitrification reaction: pyridine is reacted with concentrated nitric acid, and the reaction product is fluorinated with hydrogen fluoride gas to obtain 3-fluoro-5-nitropyridine.
-Through nitric acid esterification reaction: reacting pyridine with nitrate ester, and then fluorinating the reaction product with hydrogen fluoride gas, 3-fluoro-5-nitropyridine can be prepared.
Safety Information:
-3-fluoro-5-nitropyridine is an organic compound, which has high toxicity and explosiveness and should be used with care.
-Avoid contact with skin, eyes and respiratory tract, and wear protective equipment.
-Keep away from fire and heat sources during use and storage, and avoid severe impact and friction.
Please note that when conducting chemical experiments or preparing compounds, correct operating procedures should be followed and laboratory safety standards should be strictly observed.
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: 3-Fluoro-5-nitropyridine Visit Supplier Webpage Request for quotationCAS: 1060804-39-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 3-Fluoro-5-nitropyridine Request for quotation
CAS: 1060804-39-8
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 1060804-39-8
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Product Name: 3-Fluoro-5-nitropyridine Visit Supplier Webpage Request for quotation
CAS: 1060804-39-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 1060804-39-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 3-Fluoro-5-nitropyridine Visit Supplier Webpage Request for quotationCAS: 1060804-39-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 3-Fluoro-5-nitropyridine Request for quotation
CAS: 1060804-39-8
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 1060804-39-8
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Product Name: 3-Fluoro-5-nitropyridine Visit Supplier Webpage Request for quotation
CAS: 1060804-39-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 1060804-39-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


