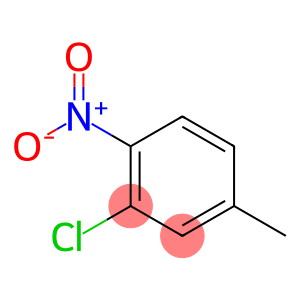
3-Chloro-4-nitrotoluene
3-Chloro-4-nitrotoluene
CAS: 38939-88-7
Molecular Formula: C7H6ClNO2
3-Chloro-4-nitrotoluene - Names and Identifiers
| Name | 3-Chloro-4-nitrotoluene |
| Synonyms | 3-Chloro-4-nitrotolu 3-Chloro-4-nitrotoluene 5-Methyl-2-nitrochlorobenzene 1-Chloro-2-nitro-5-methylbenzene 2-Chloro-4-methyl-1-nitrobenzene 1-Chloro-5-methyl-2-nitrobenzene 2-chloro-4-methyl-1-nitrobenzene Benzene, 2-chloro-4-methyl-1-nitro- |
| CAS | 38939-88-7 |
| EINECS | 254-199-6 |
| InChI | InChI=1/C7H6ClNO2/c1-5-2-3-7(9(10)11)6(8)4-5/h2-4H,1H3 |
| InChIKey | KGSQRFPDZCBVBS-UHFFFAOYSA-N |
3-Chloro-4-nitrotoluene - Physico-chemical Properties
| Molecular Formula | C7H6ClNO2 |
| Molar Mass | 171.58 |
| Density | 1.3246 (rough estimate) |
| Melting Point | 24-28 °C (lit.) |
| Boling Point | 219 °C (lit.) |
| Flash Point | >230°F |
| Vapor Presure | 0.00962mmHg at 25°C |
| Appearance | Liquid After Melting |
| Color | Clear yellow |
| Storage Condition | Sealed in dry,Room Temperature |
| Refractive Index | n20/D 1.564(lit.) |
| Physical and Chemical Properties | Yellow oily liquid |
3-Chloro-4-nitrotoluene - Risk and Safety
| Risk Codes | R22 - Harmful if swallowed R36/37/38 - Irritating to eyes, respiratory system and skin. R20/21/22 - Harmful by inhalation, in contact with skin and if swallowed. R52/53 - Harmful to aquatic organisms, may cause long-term adverse effects in the aquatic environment. |
| Safety Description | S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S61 - Avoid release to the environment. Refer to special instructions / safety data sheets. S24/25 - Avoid contact with skin and eyes. |
| UN IDs | UN 3457 6.1/PG 3 |
| WGK Germany | 3 |
| HS Code | 29049090 |
| Hazard Note | Irritant |
| Hazard Class | 6.1 |
| Packing Group | III |
3-Chloro-4-nitrotoluene - Reference Information
| NIST chemical information | Information provided by: webbook.nist.gov (external link) |
| Introduction | 3-chloro-4-nitrotoluene, also called 2-nitro-5-methylchlorobenzene, is an aryl halide. The introduction of halogen groups (-I,-Br,-Cl,-F, etc.) on aromatic compounds can significantly change the physical, chemical and biological properties of aromatic compounds. Aryl halides are not only important structural units of chemical products such as drugs, pigments, liquid crystal materials and many natural compounds, but also aryl halides are often used as raw materials for organic synthesis reactions to construct C- C bonds and C- heteroatom bonds. |
| application | 3-chloro -4-nitrotoluene can be used as organic synthesis intermediates and pharmaceutical intermediates, mainly used in laboratory research and development processes and chemical production processes. |
| preparation | silver sulfate 6.2mg, copper acetate 36.3mg, 2,9-dimethyl -1,10-phenanthroline 12.5mg, 2-nitro -5-methyl benzoic acid 36.2mg, sodium chloride 17.5mg and 4mL dimethyl sulfoxide were added to the sirek reaction tube equipped with magnetic stirrer. Heat at 160 ℃ for 24 hours in the presence of oxygen. After the reaction is completed, distilled water is added to quench the reaction, and ethyl acetate is used for extraction for 3 times, 10mL each time. The combined organic phase is concentrated to obtain 3-chloro -4-nitrotoluene 14.8mg with a yield of 43%. |
Last Update:2024-04-09 21:04:16
Supplier List
Spot supply
Product Name: 2,4-Dichlorobenzyl Alcohol Impurity 59 Visit Supplier Webpage Request for quotationCAS: 38939-88-7
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Spot supply
Product Name: 3-Chloro-4-nitrotoluene Visit Supplier Webpage Request for quotationCAS: 38939-88-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 3-Chloro-4-nitrotoluene Request for quotation
CAS: 38939-88-7
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 38939-88-7
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Spot supply
Product Name: 2,4-Dichlorobenzyl Alcohol Impurity 59 Visit Supplier Webpage Request for quotationCAS: 38939-88-7
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Spot supply
Product Name: 3-Chloro-4-nitrotoluene Visit Supplier Webpage Request for quotationCAS: 38939-88-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 3-Chloro-4-nitrotoluene Request for quotation
CAS: 38939-88-7
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 38939-88-7
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
View History


