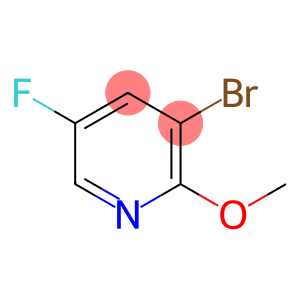
3-Bromo-5-Fluoro-2-Methoxypyridine
3-Bromo-5-Fluoro-2-Methoxypyridine
CAS: 884494-81-9
Molecular Formula: C6H5BrFNO
3-Bromo-5-Fluoro-2-Methoxypyridine - Names and Identifiers
| Name | 3-Bromo-5-Fluoro-2-Methoxypyridine |
| Synonyms | uoro-2-methoxypyridine 3-Bromo-5-fluoro-2-methoxypyrdine 3-Bromo-5-Fluoro-2-Methoxypyridine 3-BROMO-5-FLUORO-2-METHOXYPYRIDINE Pyridine, 3-bromo-5-fluoro-2-methoxy- pyridine, 3-bromo-5-fluoro-2-methoxy- 3-Bromo-5-fluoropyridin-2-yl methyl ether |
| CAS | 884494-81-9 |
| InChI | InChI=1/C6H5BrFNO/c1-10-6-5(7)2-4(8)3-9-6/h2-3H,1H3 |
3-Bromo-5-Fluoro-2-Methoxypyridine - Physico-chemical Properties
| Molecular Formula | C6H5BrFNO |
| Molar Mass | 206.01 |
| Density | 1.621±0.06 g/cm3(Predicted) |
| Boling Point | 188.4±35.0 °C(Predicted) |
| Flash Point | 67.715°C |
| Vapor Presure | 0.83mmHg at 25°C |
| Appearance | Liquid |
| pKa | -1.38±0.20(Predicted) |
| Storage Condition | under inert gas (nitrogen or Argon) at 2-8°C |
| Sensitive | Light Sensitive |
| Refractive Index | 1.5390 to 1.5430 |
| Physical and Chemical Properties | 2-methoxy-3-bromo-5-fluoropyridine is liquid at normal temperature and pressure with a density of 1.6g/cm3, A boiling point of 188.4±35.0°C(760 mmH) at one atmospheric pressure, the bromine atom on the pyridine ring can be reacted with the aryl boron reagent under coupling conditions to attach two aryl groups at position 3, the bromine atom on the pyridine ring can also be converted into a boron unit by reacting with triisopropyl borate under the action of n-butyl lithium. In addition, due to the electron-deficient nature of the pyridine ring, the bromine atom on the pyridine ring can be attacked by the nucleophile to obtain the product of debromination. |
3-Bromo-5-Fluoro-2-Methoxypyridine - Reference Information
| Use | 2-methoxy-3-bromo-5-fluoropyridine belongs to pyridine compounds, can be used for the synthesis of multi-substituted pyridine derivatives, mainly used in organic synthesis and Pharmaceutical Chemistry intermediates. |
| synthetic method | add 2-methoxy-5-fluoropyridine and N-bromosuccinimide (NBS) to a dry reaction flask, the air in the reaction flask was replaced with nitrogen gas, and then anhydrous tetrahydrofuran solvent was slowly added to the reaction flask, and the reaction mixture was dissolved by stirring at room temperature, the reaction mixture was heated slowly and allowed to stir at 70 degrees for several hours and 5 hours. After the reaction was completed, the reaction mixture was cooled to room temperature and the reaction mixture was filtered, the filtrate can be concentrated to obtain the target product molecule 2-methoxy-3-bromo-5-fluoropyridine. Figure 2-methoxy-3-bromo-5-fluoropyridine synthesis |
Last Update:2024-04-09 20:52:54
Supplier List
Spot supply
Product Name: 3-Bromo-5-fluoro-2-methoxypyridine Visit Supplier Webpage Request for quotationCAS: 884494-81-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 3-BROMO-5-FLUORO-2-METHOXYPYRIDINE Request for quotation
CAS: 884494-81-9
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 884494-81-9
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: 3-Bromo-5-Fluoro-2-Methoxypyridine Visit Supplier Webpage Request for quotation
CAS: 884494-81-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 884494-81-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 3-Bromo-5-fluoro-2-methoxypyridine Visit Supplier Webpage Request for quotationCAS: 884494-81-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 3-BROMO-5-FLUORO-2-METHOXYPYRIDINE Request for quotation
CAS: 884494-81-9
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 884494-81-9
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: 3-Bromo-5-Fluoro-2-Methoxypyridine Visit Supplier Webpage Request for quotation
CAS: 884494-81-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 884494-81-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


