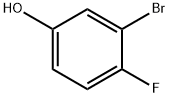
3-Bromo-4-fluorophenol
3-Bromo-4-fluorophenol
CAS: 27407-11-0
Molecular Formula: C6H4BrFO
3-Bromo-4-fluorophenol - Names and Identifiers
| Name | 3-Bromo-4-fluorophenol |
| Synonyms | 3-Bromo-4-fluorophen 3-Bromo-4-fluorophenol 3-BROMO-4-FLUOROPHENOL Phenol, 3-bromo-4-fluoro- |
| CAS | 27407-11-0 |
| EINECS | 807-954-4 |
| InChIKey | QWTULQLVGNZMLF-UHFFFAOYSA-N |
3-Bromo-4-fluorophenol - Physico-chemical Properties
| Molecular Formula | C6H4BrFO |
| Molar Mass | 191 |
| Density | 1.764±0.06 g/cm3(Predicted) |
| Boling Point | 78°C/0.5mmHg(lit.) |
| Appearance | Crystalline |
| Color | Yellow-green |
| pKa | 9.01±0.18(Predicted) |
| Storage Condition | Inert atmosphere,2-8°C |
3-Bromo-4-fluorophenol - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| Hazard Note | Irritant |
| Hazard Class | 6.1 |
3-Bromo-4-fluorophenol - Reference Information
| introduction | 3-bromo-4-fluorophenol is a yellow-green needle-like solid at normal temperature and pressure. it is a phenol derivative with certain acidity and special smell of phenol. 3-Bromo-4-fluorophenol is an important raw material for the production of certain resins, fungicides, preservatives and drug molecules. |
| uses | 3-bromo-4-fluorophenol is often used as a synthetic intermediate for drug molecules and bioactive substances, which is widely used in the preparation of fungicides and preservatives. In the synthetic transformation, it mainly revolves around the bromine unit in its structure. Bromine atoms can be connected to aryl groups, alkyl groups, etc. through Suzuki coupling reactions. In addition, phenolic hydroxyl keyi undergo alkylation under alkaline conditions to obtain ether compounds. |
| synthesis method | under the condition of ice water bath ( 0°C), 3-bromo -4-fluorophenol (24.4g, 80 mmol) protected by dimethyl tert-butylsilane is added to the solution in THF (160 mL) THF (80 mL, 80 mmol) solution, stir the mixture at 0 ℃ for 0.5 hours, concentrate the mixture to remove tetrahydrofuran solvent and unreacted tetrabutylammonium fluoride, dilute the mixture with ethyl acetate/n-hexane (1:1), then wash with water (3x) and brine, separate the organic layer, dry the organic layer with anhydrous sodium sulfate, filter out anhydrous sodium sulfate solid, and concentrate the obtained filtrate, dissolve the crude product in n-hexane (if it is insoluble, it can be heated to dissolve it), and separate and purify the residue by column chromatography (elute with a gradient of 0 to 50% ethyl acetate/hexane) to obtain a light yellow The oily target product (12.5 g,65.4 mmol,82% yield). |
Last Update:2024-04-09 21:11:58
Supplier List
Spot supply
Product Name: 3-Bromo-4-fluorophenol Visit Supplier Webpage Request for quotationCAS: 27407-11-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 3-Bromo-4-fluorophenol Request for quotation
CAS: 27407-11-0
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 27407-11-0
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: 3-Bromo-4-fluorophenol Request for quotation
CAS: 27407-11-0
Tel: 0086-13605431940
Email: xuli678323@126.com
Mobile: +86-13605431940
QQ: 56860987
Wechat: 13605431940
WhatsApp: 13605431940
CAS: 27407-11-0
Tel: 0086-13605431940
Email: xuli678323@126.com
Mobile: +86-13605431940
QQ: 56860987
Wechat: 13605431940
WhatsApp: 13605431940
Spot supply
Product Name: 3-Bromo-4-fluorophenol Visit Supplier Webpage Request for quotationCAS: 27407-11-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 3-Bromo-4-fluorophenol Request for quotation
CAS: 27407-11-0
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 27407-11-0
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: 3-Bromo-4-fluorophenol Request for quotation
CAS: 27407-11-0
Tel: 0086-13605431940
Email: xuli678323@126.com
Mobile: +86-13605431940
QQ: 56860987
Wechat: 13605431940
WhatsApp: 13605431940
CAS: 27407-11-0
Tel: 0086-13605431940
Email: xuli678323@126.com
Mobile: +86-13605431940
QQ: 56860987
Wechat: 13605431940
WhatsApp: 13605431940
View History


