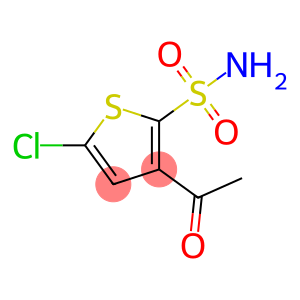
3-Acetyl-5-chlorothiophene-2-sulfonamide
3-Acetyl-5-chlorothiophene-2-sulfonamide
CAS: 160982-10-5
Molecular Formula: C6H6ClNO3S2
3-Acetyl-5-chlorothiophene-2-sulfonamide - Names and Identifiers
| Name | 3-Acetyl-5-chlorothiophene-2-sulfonamide |
| Synonyms | Metomidine hydrochloride 3-Acetyl-5-chlorothiophene-2-sulfonamide 2-Thiophenesulfonamide,3-Acetyl-5-Chloro- 2-thiophenesulfonamide, 3-acetyl-5-chloro- 2-Thiophenesulfonamide, 3-Acetyl-5-Chloro- 3-Acetyl-2-(aMinosulfonyl)-5-chlorothiophene 3-Acetyl-5-chloro-2-thiophenesulfonamide(For export only) |
| CAS | 160982-10-5 |
| EINECS | 605-240-5 |
| InChI | InChI=1/C6H6ClNO3S2/c1-3(9)4-2-5(7)12-6(4)13(8,10)11/h2H,1H3,(H2,8,10,11) |
3-Acetyl-5-chlorothiophene-2-sulfonamide - Physico-chemical Properties
| Molecular Formula | C6H6ClNO3S2 |
| Molar Mass | 239.7 |
| Density | 1.583 |
| Melting Point | 182 °C(dec.) |
| Boling Point | 453.0±55.0 °C(Predicted) |
| Flash Point | 227.7°C |
| Solubility | soluble in Methanol |
| Vapor Presure | 2.15E-08mmHg at 25°C |
| Appearance | powder to crystal |
| Color | White to Light yellow |
| pKa | 9.29±0.60(Predicted) |
| Storage Condition | under inert gas (nitrogen or Argon) at 2–8 °C |
| Refractive Index | 1.596 |
3-Acetyl-5-chlorothiophene-2-sulfonamide - Reference Information
| introduction | 3-acetyl -5-chlorothiophene-2-sulfonamide is an intermediate for the preparation of brinzolamide. brinzolamide is a new type of local carbonic anhydrase inhibitor. it is a heterocyclic sulfonamide drug and was approved for marketing in the United States in 1998. Brinzolamide has a high degree of selectivity and affinity, and significantly inhibits the activity of carbonic anhydrase, effectively reducing intraocular pressure. Brinzolamide has a long half-life, less irritation to the eye, its physiological pH and suspension design make the eye dripping comfortable, tolerable and safe, and less adverse reactions (usually without treatment It can relieve itself), is a very valuable new anti-glaucoma drug. |
| prepare | 5L four-mouth bottle with 210 g3-acetyl -5-chloro -2-(benzyl mercapto) thiophene, 1500g acetonitrile, stirring, 320g glacial acetic acid and 200g water, controlling the temperature between 0-15 ℃, adding 241.6g trichloroisocyanuric acid in batches, adding about 30min, holding the reaction at 0-15 ℃, TLC monitoring the reaction until the raw materials disappear. Water bath at 35-45 ℃, concentration under reduced pressure to remove solvent, 1500g of 25% ethyl acetate petroleum ether solution was added, stirred, filtered, and the filtrate was evaporated under reduced pressure to obtain 182.7g of light yellow oil, I .e. 3-acetyl -5-chloro -2-thiophene sulfonyl chloride. The above sulfonyl chloride is diluted with 105g of ethyl acetate, controlled at 0~15 ℃, added dropwise to 420g of ammonia water, stirred and reacted until the sulfonyl chloride disappears, cooled to about 0 ℃, stirred and precipitated for 1h. Filtration, a small amount of blistering and washing, the obtained solid is dried at 40 ℃ to obtain 162.4g of white-like 3-acetyl -5-chlorothiophene-2-sulfonamide with a yield of 91.2%. HPLC purity is greater than 99%. |
Last Update:2024-04-09 21:01:54
Supplier List
Spot supply
Product Name: 3-Acetyl-5-chlorothiophene-2-sulfonamide Visit Supplier Webpage Request for quotationCAS: 160982-10-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 3-Acetyl-5-chlorothiophene-2-sulfonamide Request for quotation
CAS: 160982-10-5
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 160982-10-5
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Product Name: 3-Acetyl-5-chloro-2-thiophenesulfonamide Visit Supplier Webpage Request for quotation
CAS: 160982-10-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 160982-10-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 3-Acetyl-5-chlorothiophene-2-sulfonamide Visit Supplier Webpage Request for quotationCAS: 160982-10-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 3-Acetyl-5-chlorothiophene-2-sulfonamide Request for quotation
CAS: 160982-10-5
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 160982-10-5
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Product Name: 3-Acetyl-5-chloro-2-thiophenesulfonamide Visit Supplier Webpage Request for quotation
CAS: 160982-10-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 160982-10-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


