29560-58-5
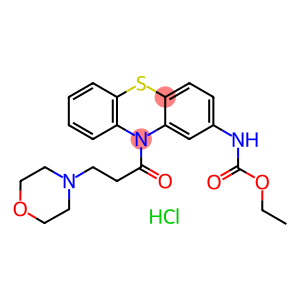
Moricizine Hydrochloride
CAS: 29560-58-5
Molecular Formula: C22H26ClN3O4S
29560-58-5 - Names and Identifiers
29560-58-5 - Physico-chemical Properties
| Molecular Formula | C22H26ClN3O4S |
| Molar Mass | 463.98 |
| Melting Point | 189° (dec) |
| Boling Point | 625°C at 760 mmHg |
| Flash Point | 331.8°C |
| Vapor Presure | 1.54E-15mmHg at 25°C |
| Storage Condition | Room Temprature |
29560-58-5 - Risk and Safety
| HS Code | 2934302700 |
| Toxicity | LD50 in mice, rats (mg/kg): 36, 12 i.v.; in mice (mg/kg): 131 i.p. (Kaverina) |
29560-58-5 - Nature
Open Data Verified Data
white crystals were obtained from dichloroethane. Soluble in water or alcohol. Melting point 189 °c (decomposition).
29560-58-5 - Preparation Method
Open Data Verified Data
In the solution of phenothiazine-2-carbamic acid ethyl ester in anhydrous toluene, add 3 chloro propionyl chloride Dropwise, Reflux reaction, add activated carbon for hot filtration, treatment gave 10-(3-chloropropionyl) phenothiole-2-carbamic acid ethyl ester. The compound was dissolved in toluene and morpholine was added to the reaction. Moricizine was obtained by post-treatment. Moricizine was extracted with toluene, and the extract was dried over anhydrous magnesium sulfate. To the anhydrous toluene solution, a solution of hydrogen chloride in anhydrous ether was added to precipitate moricizine hydrochloride, which was then recrystallized from methylene chloride to give moricizine hydrochloride.
29560-58-5 - Standard
Authoritative Data Verified Data
This product is 10-(3-morpholinopropionyl) phenothiazine-2-carbamic acid ethyl ester hydrochloride. The content of C22H25N304S • HCl shall not be less than 99.0% calculated as dry product.
29560-58-5 - Trait
Authoritative Data Verified Data
- This product is white or off-white powder.
- This product is soluble in methanol, soluble in water or ethanol, slightly soluble in acetone, insoluble in ethyl acetate; Soluble in glacial acetic acid.
absorption coefficient
take the right amount of this product, precision weighing, plus ethanol-water (1:1) dissolved and quantitative dilution made in each lml containing about 0.lmg solution; Take appropriate amount of precision, dilute with water to make a solution containing about 10ug per lml, and measure absorbance at 268mn according to UV-visible spectrophotometry (General rule 0401), the absorption coefficient was 360 to 375 as calculated from the dried product.
29560-58-5 - Use
Open Data Verified Data
U. S. Du pert company developed. Listed in 1990. Antiarrhythmic drugs. Phenothiazine derivatives, sodium channel blockers, and have local anesthetic activity. With a moderate dilating coronary, spasmolytic and anti-M-cholinergic effect, also has a pronounced anti-tachyarrhythmia effect, the effect of which is similar to quinidine. For atrial and ventricular premature beat, paroxysmal tachycardia, atrial fibrillation and atrial flutter has a significant effect.
29560-58-5 - Differential diagnosis
Authoritative Data Verified Data
- take about 10mg of this product, add 5ml of water to dissolve, add 1 drop of bismuth potassium iodide solution, and immediately produce orange red precipitate.
- take about 15mg of this product, add 2ml of water to dissolve, add 1 mol/L hydroxylamine hydrochloride solution 0.5ml, Dropwise add 5mol/L ethanol to make potassium hydroxide solution alkaline, forming white precipitate, the mixture was heated and boiled for a few minutes, cooled, and the mixture was made acidic by adding dilute hydrochloric acid. The mixture was turned into light purple, and 1-2 drops of ferric chloride solution were added dropwise to turn into deep purple.
- The infrared absorption spectrum of this product should be consistent with that of the control (Spectrum set 651).
- the aqueous solution of this product was chloride identification (1) of the reaction (General 0301).
29560-58-5 - Safety
Open Data Verified Data
mice, rats were injected intravenously with LDso:36.12 mg/kg. Mice were injected intraperitoneally with LDso:131mg/kg.
29560-58-5 - Exam
Authoritative Data Verified Data
Related substances
take 25mg of this product, put it in a 25ml measuring flask, add 0.02mol/L hydrochloric acid solution-acetonitrile (58:42) mixed solution to dissolve and dilute to the scale, shake, as a test solution, 1ml was accurately weighed, placed in a 100ml measuring flask, diluted to a scale with the above mixed solution, and shaken to obtain a control solution. Tested according to high performance liquid chromatography (General 0512). The mobile phase consisted of acetonitrile-water-triethylamine (420:580:1) containing 0.003mol/L sodium octane sulfonate and pH adjusted to 4.2 with glacial acetic acid; the detection wavelength was 270nm; The column temperature was 35°C. The number of theoretical plates shall not be less than 3000 calculated by the peak of moricizine, and the separation degree between the peak of moricizine and the adjacent impurity peaks shall meet the requirements. Accurately take 20 u1 of the test solution and the control solution, respectively inject human liquid chromatography, record the chromatogram to 5.5 times the retention time of the main component peak, if there are impurity peaks in the chromatogram of the test solution, the area of a single impurity peak shall not be greater than 0.5 times (0.5%) of the area of the main peak of the control solution; the sum of each impurity peak area shall not be greater than the main peak area of the control solution (1.0%).
loss on drying
take this product, dry to constant weight at 105°C, weight loss shall not exceed 1.0% (General rule 0831).
ignition residue
take 1.0g of this product and check it according to law (General rule 0841). The remaining residue shall not exceed 0.1%.
Heavy metals
The residue left under the item of taking the ignition residue shall not contain more than 20 parts per million of heavy metal when examined by law (General rule 0821, Law II).
29560-58-5 - Content determination
Authoritative Data Verified Data
take this product about 0.35g, precision weighing, add glacial acetic acid 10ml dissolved, add acetic anhydride 30ml, according to the potential titration method (General rule 0701), with high gas acid titration solution (0.1 mol/L) titration, and the titration results were corrected with a blank test. Each 1 ml of high gas acid titrant (0.1 mol/L) corresponds to 46.40mg of C22H25N304S • HCl.
29560-58-5 - Category
Authoritative Data Verified Data
antiarrhythmic drugs.
29560-58-5 - Storage
Authoritative Data Verified Data
light shielding, sealed storage.
29560-58-5 - Morexizide hydrochloride tablets
Authoritative Data Verified Data
This product contains moricizine hydrochloride (C22H25N304S • HCl) should be 90.0% ~ 110.0% of the label.
trait
This product is sugar-coated tablet or film-coated tablet, white or almost White after removing the coating.
identification
- take this product, remove the coating, triturate, take an appropriate amount (approximately equivalent to 15mg of moricizine hydrochloride), add 2ml of water, shake to dissolve moricepine hydrochloride, filter, the filtrate was added with 0.5ml of 1 mol/L hydroxylamine hydrochloride solution and 5mol/L ethanol to make potassium hydroxide solution alkaline and white precipitate. The mixture was heated and boiled for several minutes, cooled, and diluted hydrochloric acid was added to make it acidic, the mixture turned pale purple, and 1 to 2 drops of ferric chloride solution were added dropwise to turn deep purple.
- take this product, remove the coating, triturate, weigh an appropriate amount (approximately equivalent to 30mg of moricizine hydrochloride), add 10ml of water, shake to dissolve moricizine hydrochloride, filter, the filtrate shows the reaction of chloride identification (1) (General 0301).
examination
- for related substances, take an appropriate amount of fine powder under the content determination item (about 50mg equivalent to moricizine hydrochloride), put it in a 50ml measuring flask, and add 0.02mol/L hydrochloric acid solution-acetonitrile (58:42) Mix the appropriate amount of solution, shake to dissolve moricizine hydrochloride and dilute to the scale, shake, filter, and take the filtrate as the test solution; Take 1ml, in a 100ml measuring flask, the mixture was diluted to the mark with the above mixed solution, and the mixture was shaken to obtain a control solution. If there are impurity peaks in the chromatogram of the test solution, the single impurity peak area shall not be greater than the main peak area of the control solution (1.0% ) ; the sum of each impurity peak area shall not be greater than 1.5 times (1.5%) of the main peak area of the control solution.
- dissolution of this product, according to the dissolution and release determination method (General 0931 first method), with 0.lmol/L hydrochloric acid solution 1000ml as the dissolution medium, the speed is 50 rpm, according to the operation, after 60 minutes, take the solution, filtering, precision take the appropriate amount of filtrate, A solution containing about 10ug moricizine hydrochloride per 1 ml was prepared by quantitative dilution with dissolution medium. According to UV-visible spectrophotometry (General rule 0401), the absorbance is measured at the wavelength of 268mn, and the dissolution of each tablet is calculated according to the absorption coefficient of C22H25N304S • HC1 as 367, the limit is 80% of the labeled amount and shall be in accordance with the provisions.
- others shall be in accordance with the relevant provisions under the item of tablets (General rule 0101).
Content determination
Take 20 tablets of this product, remove the coating, precision weighing, fine, precision weighing appropriate amount (about 50mg moricizine hydrochloride), put in 250ml measuring flask, add 25ml of ethanol, fully shake to dissolve moricizine hydrochloride, dilute to scale with water, shake well, filter, take 5ml of continued filtrate accurately, put it in 100ml measuring flask, dilute with water to the scale, shake, according to UV-visible spectrophotometry (General 0401), at the wavelength of 268nm absorbance, calculated as C22H25N304S • HC1 absorption coefficient of 367, that's right.
category
Same as moricizine hydrochloride.
specification
50mg
storage
light shielding, sealed storage.
Supplier List
CAS: 29560-58-5
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 29560-58-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 29560-58-5
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 29560-58-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025

