286930-02-7
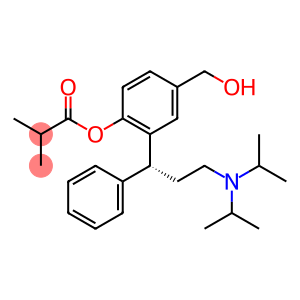
2-Methylpropionic acid 2-[3-(N,N-diisopropylamino)-1(R)-phenylpropyl]-4-(hydroxymethyl)phenyl ester
CAS: 286930-02-7
Molecular Formula: C26H37NO3
286930-02-7 - Names and Identifiers
286930-02-7 - Physico-chemical Properties
| Molecular Formula | C26H37NO3 |
| Molar Mass | 411.58 |
| Density | 1.043 |
| Storage Condition | 2-8℃ |
| In vitro study | Fesoterodine decreases micturition frequency, urgency severity and urgency incontinence episodes and increases the volume voided with each micturition. After oral administration, Fesoterodine is rapidly and extensively hydrolysed in plasma by nonspecific esterases to Desfesoterodine (5-hydroxymethyl tolterodine; SPM 7605; HY-76569; an active metabolite of Fesoterodine). |
286930-02-7 - Preparation solution concentration reference
| 1mg | 5mg | 10mg | |
|---|---|---|---|
| 1 mM | 2.43 ml | 12.148 ml | 24.297 ml |
| 5 mM | 0.486 ml | 2.43 ml | 4.859 ml |
| 10 mM | 0.243 ml | 1.215 ml | 2.43 ml |
| 5 mM | 0.049 ml | 0.243 ml | 0.486 ml |
Last Update:2024-01-02 23:10:35
286930-02-7 - Reference Information
| overview | frostidine (fesoterodine, trade name ToviazTM) is a new type of m receptor blocker, which is rapidly metabolized into active metabolites in the body and plays a role. |
| use | Frostine was approved by FDA on October 31, 2008 for the treatment of overactive bladder (overactivebladder,OAB). |
| Biological activity | Fesoterodine is an orally effective, non-subtype selective, competitive muscarinic receptor (mAChR) antagonist. The pKi values for M1,M2,M3,M4,M5 receptors are 8.0,7.7,7.4,7.3,7.5, respectively. Fesoterodine for overactive bladder (OAB). |
| target | pKi: 8.0 (M1), 7.7 (M2), 7.4 (M3), 7.3 (M4) and 7.5 (M5) |
| Animal Model: | Bladders from female Sprague-Dawley rats (225-275 g) |
| Dosage: | 0.01, 0.1 and 1 mg/kg |
| Administration: | IV |
| Result: | Reduced the micturition pressure and increased bladder capacity and ICIs at the lowest dose tested of 0.01 mg/kg. |
Last Update:2024-04-09 15:16:49
Supplier List
Spot supply
Product Name: (R) Fesoterodine Visit Supplier Webpage Request for quotationCAS: 286930-02-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: (R) Fesoterodine Request for quotation
CAS: 286930-02-7
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 286930-02-7
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: (R) Fesoterodine Visit Supplier Webpage Request for quotation
CAS: 286930-02-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 286930-02-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: (R) Fesoterodine Visit Supplier Webpage Request for quotationCAS: 286930-02-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: (R) Fesoterodine Request for quotation
CAS: 286930-02-7
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 286930-02-7
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: (R) Fesoterodine Visit Supplier Webpage Request for quotation
CAS: 286930-02-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 286930-02-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


