28300-74-5
Antimony potassium tartrate trihydrate
CAS: 28300-74-5
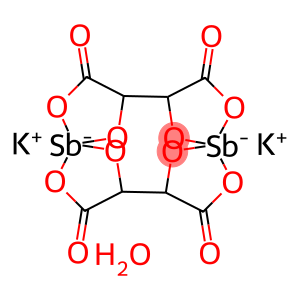
Molecular Formula: C8H4O12Sb2.3H2O.2K
28300-74-5 - Names and Identifiers
| Name | Antimony potassium tartrate trihydrate |
| Synonyms | Potassium antimony t Antimony potassium tartrate Antimony potassium tartrate powder Antimony potassium tartrate trihydrate POTASSIUM ANTIMONY(III) OXIDE TARTRATE H Potassium antimony(III) oxide tartrate trihydrate ANTIMONY POTASSIUM TARTRATE, SOLUTIONANTIMONY POTASSIUM TARTRATE, SOLUTIONANTIMONY POTASSIUM TARTRATE, SOLUTION ANTIMONY POTASSIUM TARTRATE, TRIHYDRATE, REAGENT (ACS)ANTIMONY POTASSIUM TARTRATE, TRIHYDRATE, REAGENT (ACS)ANTIMONY POTASSIUM TARTRATE, TRIHYDRATE, REAGENT (ACS) |
| CAS | 28300-74-5 |
| EINECS | 608-190-2 |
| InChI | InChI=1/2C4H4O6.2K.H2O.2Sb/c2*5-1(3(7)8)2(6)4(9)10;;;;;/h2*1-2H,(H,7,8)(H,9,10);;;1H2;;/q2*-2;2*+1;;2*+3/p-4/rC8H6O12Sb2.2K.H2O/c9-5(10)1-3-7(13)19-22(17-3)16-2(6(11)12)4-8(14)20-21(15-1)18-4;;;/h1-4H,(H,9,10)(H,11,12);;;1H2/q;2*+1;/p-2 |
| InChIKey | BBUFTTIHMIIQLI-UHFFFAOYSA-J |
28300-74-5 - Physico-chemical Properties
| Molecular Formula | C8H4O12Sb2.3H2O.2K |
| Molar Mass | 667.87 |
| Density | 2,607 g/cm3 |
| Melting Point | ≥300°C(lit.) |
| Specific Rotation(α) | D20 +140.69° (c = 2 in water), +139.25° (c = 2 in glycerol) |
| Water Solubility | 8.3 g/100 mL |
| Solubility | water: soluble |
| Appearance | Powder |
| Specific Gravity | 2.6 |
| Color | White |
| Exposure Limit | ACGIH: TWA 0.5 mg/m3NIOSH: IDLH 50 mg/m3; TWA 0.5 mg/m3 |
| Merck | 14,700 |
| PH | 4.0 (50g/l, H2O, 20℃) |
| Storage Condition | Store at +15°C to +25°C. |
| Stability | Stable. Incompatible with alkalies, carbonates, strong oxidizing agents. |
| Physical and Chemical Properties | Colorless transparent crystal or white crystalline powder, odorless, sweet. The relative density was 2.607. Soluble in water (8.3g/100mL,25 °c; 35.9g/100mL,100 °c) and glycerol, insoluble in ethanol. In the air will be weathered, the aqueous solution is slightly acidic. |
| Use | This product is used as a mordant for dyeing cotton fabrics with alkaline dyes. After dyeing with acidic dyes, treating with this product can improve the washing and soaping fastness of the fabric. It can also be used as a leather mordant. |
28300-74-5 - Risk and Safety
| Risk Codes | R20/22 - Harmful by inhalation and if swallowed. R51/53 - Toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment. |
| Safety Description | 61 - Avoid release to the environment. Refer to special instructions / safety data sheets. |
| UN IDs | UN 1551 6.1/PG 3 |
| WGK Germany | 3 |
| RTECS | CC6825000 |
| TSCA | Yes |
| HS Code | 29181300 |
| Hazard Class | 6.1 |
| Packing Group | III |
| Toxicity | LD50 in mice (mg/kg): 55 s.c.; 65 i.v. (Ercoli) |
28300-74-5 - Reference Information
| EPA chemical information | Information provided by: ofmpub.epa.gov (external link) |
| use | as an anti-bloodsucking medicine added to animal feed. in Japan, the 90% of this product is used as a fixing agent for dyes, I .e. as a fixing agent for basic dyes such as cotton, skin, sheep, etc. and as an acid dye for nylon. The remaining 10% is used as a fading inhibitor for polyvinyl chloride, as well as for the manufacture of pigments and pesticide impurities. Antimony potassium tartrate, as an anti-schistosomiasis drug, is a variety included in my country's national essential drug list. Intravenous injection of antimony potassium tartrate can directly cause changes in the physiological function and tissue morphology of schistosomiasis, which is manifested as the worm body curls up, the suction cup loses the ability to suck the blood vessel wall, and is brought into the liver. Schistosoma was repeatedly treated with antimony agent, the body atrophy, reproductive organs degeneration, female stop laying eggs, and finally surrounded by host cells, forming dead insect nodules. Antimony compounds, like arsenic compounds, have corrosive effects. It can irritate the stomach, cause nausea, and cause acute gastric catarrh, which in turn causes watery diarrhea. It also has a stimulating effect on the skin and mucous membranes, especially the sweating skin itching and rash. Its side effects are similar to arsenic poisoning. Its symptoms are severe exfoliative enteritis, steatosis of the large secretory glands, weakened heart, and paralysis of the central nervous system, manifested as progressive unconsciousness and impotence paralysis. Analyze sodium, germanium, etc., mordant, insecticide. |
| Production method | is obtained from tartaric acid by antimonizing. Mix 4 parts of antimony trioxide and 5 parts of potassium tartrite hydrogen, add 40 parts of hot water, stir and heat to dissolve most of them, filter while hot, and cool and crystallize. After filtration, recrystallization is carried out. In actual operation, antimony trioxide can be obtained by the reaction of antimony trioxide and sodium carbonate, and potassium hydrogen tartrate can be obtained by tartaric acid and potassium carbonate. It is obtained by the reaction of tartaric acid with antimony trioxide and potassium carbonate: dissolve tartaric acid and antimony trioxide in hot water, add potassium carbonate to adjust the PH to 3.8, and boil for 2-3h. Then adjust the PH to 5.4-6.2 with potassium carbonate and continue boiling for 2-3 hours. After cooling, decolorize with activated carbon, and the decolorization solution is concentrated, crystallized and dried (70-80 ℃) to obtain antimony potassium tartrate. |
| category | toxic substances |
| toxicity classification | highly toxic |
| acute toxicity | oral administration-rat LD50: 115 mg/kg; Oral administration-mouse LDL0: 600 mg/kg |
| flammability hazard characteristics | combustible; combustion produces toxic potassium oxide and antimonide smoke |
| storage and transportation characteristics | warehouse ventilation and low temperature drying; separate from food raw materials storage and transportation |
| fire extinguishing agent | carbon dioxide, sand, water, foam |
| occupational standard | TWA 0.5 mg (antimony)/m3; STEL 2.5 mg (antimony)/m3 |
| toxic substance data | information provided by: pubchem.ncbi.nlm.nih.gov (external link) |
Last Update:2024-04-09 02:00:07
Supplier List
Multiple SpecificationsSpot supply
Product Name: Dipotassium bis[μ-tartrato(4-)]diantimonate(2-) trihydrate Visit Supplier Webpage Request for quotationCAS: 28300-74-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Potassium antimonyl tartrate sesquihydrate Request for quotation
CAS: 28300-74-5
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 28300-74-5
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Multiple SpecificationsSpot supply
Product Name: Dipotassium bis[μ-tartrato(4-)]diantimonate(2-) trihydrate Visit Supplier Webpage Request for quotationCAS: 28300-74-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Potassium antimonyl tartrate sesquihydrate Request for quotation
CAS: 28300-74-5
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 28300-74-5
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
View History

