274-76-0
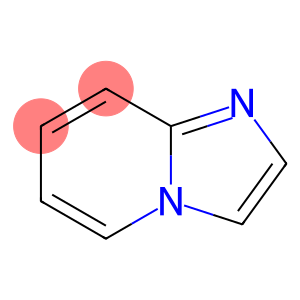
imidazo(1,2-A)pyridine
CAS: 274-76-0
Molecular Formula: C7H6N2
274-76-0 - Names and Identifiers
| Name | imidazo(1,2-A)pyridine |
| Synonyms | Pyrimidazole 1-Azaindolizine 1,3a-Diazaindene IMIDAZO[1,2-A]PYRIDINE imidazo(1,2-A)pyridine Imidazo[1,2-a]pyridine H-imidazo[1,2-a]pyridine N-[4-(4-nitrophenyl)sulfonylphenyl]-2-(1-piperidinyl)acetamide |
| CAS | 274-76-0 |
| InChI | InChI=1/C7H6N2/c1-2-5-9-6-4-8-7(9)3-1/h1-6H |
274-76-0 - Physico-chemical Properties
| Molecular Formula | C7H6N2 |
| Molar Mass | 118.14 |
| Density | 1.165g/mLat 25°C(lit.) |
| Boling Point | 103°C1mm Hg(lit.) |
| Flash Point | >230°F |
| Solubility | Chloroform, Methanol |
| Appearance | Powder |
| Color | White to off-white |
| Maximum wavelength(λmax) | ['297nm(EtOH)(lit.)'] |
| pKa | 6.80±0.30(Predicted) |
| Storage Condition | Keep in dark place,Inert atmosphere,Room temperature |
| Refractive Index | n20/D 1.626(lit.) |
| MDL | MFCD00005553 |
274-76-0 - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| Risk Codes | 36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S37/39 - Wear suitable gloves and eye/face protection |
| WGK Germany | 3 |
| HS Code | 29339900 |
| Hazard Class | IRRITANT |
274-76-0 - Introduction
imidazo (1,2-a) pyridine, also known as imidazo (1,2-a) pyridine, is a heterocyclic compound containing imidazole and pyridine rings. Its chemical formula is C8H6N2 and its molecular weight is 134.15g/mol. The following is a description of the nature, use, preparation and safety information of imidazo (1,2-a) pyridine:
Nature:
- imidazo (1,2-a) pyridine is a colorless to pale yellow crystalline solid with an odor similar to imidazole and pyridine.
-Its melting point is about 140-150 ° C. It can be dissolved in organic solvents, such as ethers, alcohols and ketones, but it is almost insoluble in water.
- imidazo (1,2-a) pyridine is a basic compound that can react with acids to form salts.
Use:
- imidazo (1,2-a) pyridine is an important organic synthesis intermediate, which is widely used in the fields of medicine, pesticide and dye.
-In the pharmaceutical industry, it is often used to synthesize antiviral drugs, anti-tumor drugs and antibacterial drugs.
-In pesticide manufacturing, it can be used as a synthetic raw material for pesticides and herbicides.
- imidazo (1,2-a) pyridine can also be used in the preparation of organic electronic devices.
Method:
- imidazo (1,2-a) pyridine has many preparation methods, one of which is commonly used by the reaction of pyridine and cyanide or cyanate ester.
-Another preparation method is to react pyridoxine aromatic amine with ketones to generate imine, and then obtain imidazo (1,2-a) pyridine through cyclization reaction.
Safety Information:
- imidazo (1,2-a) pyridine is less toxic, but care should still be taken to follow safe practices.
-It may irritate the eyes and skin, so avoid direct contact during operation.
- imidazo (1,2-a) pyridine should be stored in a cool, dry place, away from fire and oxidizing agents.
Nature:
- imidazo (1,2-a) pyridine is a colorless to pale yellow crystalline solid with an odor similar to imidazole and pyridine.
-Its melting point is about 140-150 ° C. It can be dissolved in organic solvents, such as ethers, alcohols and ketones, but it is almost insoluble in water.
- imidazo (1,2-a) pyridine is a basic compound that can react with acids to form salts.
Use:
- imidazo (1,2-a) pyridine is an important organic synthesis intermediate, which is widely used in the fields of medicine, pesticide and dye.
-In the pharmaceutical industry, it is often used to synthesize antiviral drugs, anti-tumor drugs and antibacterial drugs.
-In pesticide manufacturing, it can be used as a synthetic raw material for pesticides and herbicides.
- imidazo (1,2-a) pyridine can also be used in the preparation of organic electronic devices.
Method:
- imidazo (1,2-a) pyridine has many preparation methods, one of which is commonly used by the reaction of pyridine and cyanide or cyanate ester.
-Another preparation method is to react pyridoxine aromatic amine with ketones to generate imine, and then obtain imidazo (1,2-a) pyridine through cyclization reaction.
Safety Information:
- imidazo (1,2-a) pyridine is less toxic, but care should still be taken to follow safe practices.
-It may irritate the eyes and skin, so avoid direct contact during operation.
- imidazo (1,2-a) pyridine should be stored in a cool, dry place, away from fire and oxidizing agents.
Last Update:2024-04-09 20:49:11
Supplier List
Spot supply
Product Name: Imidazo[1,2-a]pyridine Visit Supplier Webpage Request for quotationCAS: 274-76-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Imidazo[1,2-a]pyridine Request for quotation
CAS: 274-76-0
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 274-76-0
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: Imidazo[1,2-a]Pyridine Visit Supplier Webpage Request for quotation
CAS: 274-76-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 274-76-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Imidazo[1,2-a]pyridine Visit Supplier Webpage Request for quotationCAS: 274-76-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Imidazo[1,2-a]pyridine Request for quotation
CAS: 274-76-0
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 274-76-0
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: Imidazo[1,2-a]Pyridine Visit Supplier Webpage Request for quotation
CAS: 274-76-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 274-76-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


