262297-13-2
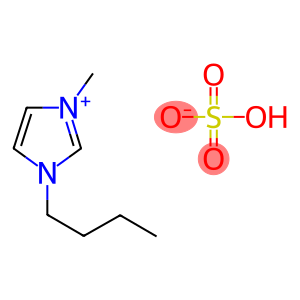
1-Butyl-3-methylimidazolium hydrogen sulfate
CAS: 262297-13-2
Molecular Formula: C8H16N2O4S
262297-13-2 - Names and Identifiers
262297-13-2 - Physico-chemical Properties
| Molecular Formula | C8H16N2O4S |
| Molar Mass | 236.29 |
| Density | 1.27g/ml |
| Melting Point | 29-32 °C |
| Flash Point | 284°C |
| Solubility | soluble in Methanol |
| Appearance | clear liquid |
| Color | Colorless to Red to Green |
| Storage Condition | Inert atmosphere,Room Temperature |
262297-13-2 - Risk and Safety
| Hazard Symbols | C - Corrosive |
| Risk Codes | 34 - Causes burns |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) |
| UN IDs | UN 3265 8/PG 2 |
| WGK Germany | 3 |
| FLUKA BRAND F CODES | 3-10 |
262297-13-2 - Reference Information
| ionic liquid | 1-butyl-3-methylimidazole bisulfate is an ionic liquid, which has the advantages of good thermal stability, non-volatilization, high conductivity, wide electrochemical window, etc. It has a good application prospect as an electrolyte in supercapacitors. Imidazole ionic liquids have low viscosity and high conductivity. Since 1-ethyl-3-methylimidazolium tetrafluoroborate (EMIBF4), imidazole ionic liquids developed rapidly. 1-Butyl-3-methylimidazole (BMI ^) ionic liquids have been extensively studied due to their low viscosity, relatively high conductivity and easy synthesis. B.Andrea et al. used 1-butyl-3-methylimidazolium hexafluorophosphate (BMIPF6) and 1-butyl3-butylimidazolium tetrafluoroborate (BMIBF4) as the electrolyte of activated carbon (AC)/polytrimethylthiophene (pMeT) hybrid capacitors. Compared with organic electrolyte (PC-EtNBF4) capacitors, ionic liquid capacitors have higher specific energy, power density and current efficiency at 60°C. |
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: 3-Butyl-1-methyl-1H-imidazol-3-ium hydrogen sulfate Visit Supplier Webpage Request for quotationCAS: 262297-13-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 1-BUTYL-3-METHYLIMIDAZOLIUM HYDROGENSULFATE Request for quotation
CAS: 262297-13-2
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 262297-13-2
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Spot supply
Product Name: 3-Butyl-1-methyl-1H-imidazol-3-ium hydrogen sulfate Visit Supplier Webpage Request for quotationCAS: 262297-13-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 1-BUTYL-3-METHYLIMIDAZOLIUM HYDROGENSULFATE Request for quotation
CAS: 262297-13-2
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 262297-13-2
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
View History

