259655-01-1
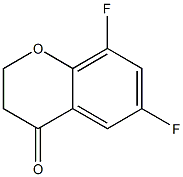
6,8-difluoro-3,4-dihydro-2H-1-benzopyran-4-one
CAS: 259655-01-1
Molecular Formula: C9H6F2O2
259655-01-1 - Names and Identifiers
259655-01-1 - Physico-chemical Properties
| Molecular Formula | C9H6F2O2 |
| Molar Mass | 184.14 |
| Density | 1.392±0.06 g/cm3(Predicted) |
| Boling Point | 282.9±40.0 °C(Predicted) |
| Storage Condition | Sealed in dry,Room Temperature |
259655-01-1 - Introduction
6,8-difluororo-3, is an organic compound with the chemical formula C12H6F2O2. The following is a description of some properties, uses, preparation and safety information about this compound:
Nature:
1. Appearance: 6,8-difluoro-3, is a solid, usually yellow crystalline powder.
2. Melting point: The melting point range of this compound is 90-93 degrees Celsius.
3. Solubility: 6,8-difluoro-3, soluble in common organic solvents, such as ethanol, methanol, dimethyl thionamide, etc.
Use:
1. Chemical synthesis: 6,8-difluoro-3, as an important organic intermediate, can be used to synthesize biologically active compounds and drugs.
2. Fluorescent chromogenic agent: Due to its special chemical structure, this compound can be used as a fluorescent dye and chromogenic agent.
Method:
6,8-difluoro-3, can be obtained by the fluorination reaction of benzopyrone derivatives, the main steps are as follows:
1. Take benzopyrone and fluorinating agent reaction, such as hydrogen fluoride gas (HF) or fluorinated sulfoxide (SO2F2), to obtain the corresponding fluorinated products.
2. The fluorine-substituted product is treated and purified under appropriate reaction conditions to finally obtain 6,8-difluoro-3, and is prepared.
Safety Information:
1. 6,8-difluoro-3, it will not cause serious harm to human body and environment under general conditions.
2. When handling and handling the compound, follow regular laboratory practices, including wearing appropriate protective equipment (such as gloves, glasses, and laboratory coats).
3. Waste disposal: Dispose of waste in accordance with local laws and regulations to avoid direct discharge or damage to the environment.
Note: When conducting any chemical experiment, please be sure to follow the correct operating procedures and pay attention to laboratory safety.
Nature:
1. Appearance: 6,8-difluoro-3, is a solid, usually yellow crystalline powder.
2. Melting point: The melting point range of this compound is 90-93 degrees Celsius.
3. Solubility: 6,8-difluoro-3, soluble in common organic solvents, such as ethanol, methanol, dimethyl thionamide, etc.
Use:
1. Chemical synthesis: 6,8-difluoro-3, as an important organic intermediate, can be used to synthesize biologically active compounds and drugs.
2. Fluorescent chromogenic agent: Due to its special chemical structure, this compound can be used as a fluorescent dye and chromogenic agent.
Method:
6,8-difluoro-3, can be obtained by the fluorination reaction of benzopyrone derivatives, the main steps are as follows:
1. Take benzopyrone and fluorinating agent reaction, such as hydrogen fluoride gas (HF) or fluorinated sulfoxide (SO2F2), to obtain the corresponding fluorinated products.
2. The fluorine-substituted product is treated and purified under appropriate reaction conditions to finally obtain 6,8-difluoro-3, and is prepared.
Safety Information:
1. 6,8-difluoro-3, it will not cause serious harm to human body and environment under general conditions.
2. When handling and handling the compound, follow regular laboratory practices, including wearing appropriate protective equipment (such as gloves, glasses, and laboratory coats).
3. Waste disposal: Dispose of waste in accordance with local laws and regulations to avoid direct discharge or damage to the environment.
Note: When conducting any chemical experiment, please be sure to follow the correct operating procedures and pay attention to laboratory safety.
Last Update:2024-04-09 21:04:16
Supplier List
Spot supply
Product Name: 6,8-Difluorochroman-4-one Visit Supplier Webpage Request for quotationCAS: 259655-01-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 6,8-Difluorochroman-4-one Request for quotation
CAS: 259655-01-1
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 259655-01-1
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: 6,8-Difluorochroman-4-one Visit Supplier Webpage Request for quotation
CAS: 259655-01-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 259655-01-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 6,8-Difluorochroman-4-one Visit Supplier Webpage Request for quotationCAS: 259655-01-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 6,8-Difluorochroman-4-one Request for quotation
CAS: 259655-01-1
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 259655-01-1
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: 6,8-Difluorochroman-4-one Visit Supplier Webpage Request for quotation
CAS: 259655-01-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 259655-01-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


