248281-84-7
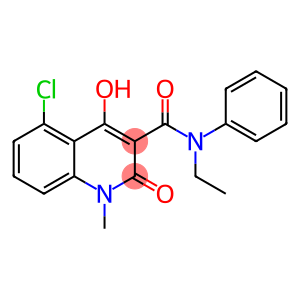
5-Chloro-N-ethyl-4-hydroxy-1-methyl-2-oxo-N-phenyl-1,2-dihydroquinoline-3-carboxamide
CAS: 248281-84-7
Molecular Formula: C19H17ClN2O3
248281-84-7 - Names and Identifiers
| Name | 5-Chloro-N-ethyl-4-hydroxy-1-methyl-2-oxo-N-phenyl-1,2-dihydroquinoline-3-carboxamide |
| Synonyms | CS-82 ABR215062 Laquinimod ABR-215062 CIVENTICHEM SAIK-MS compound CIVENTICHEM CV-4057 ABR-215062 (Laquinimod) ABR 215062 (LAQUINIMOD) LaquiniMod, SAIK-MS coMpound, ABR-215062 5-chloro-N-ethyl-2-hydroxy-1-methyl-4-oxo-N-phenyl-1,4-dihydroquinoline-3-carboxamide 5-Chloro-N-ethyl-4-hydroxy-1-methyl-2-oxo-N-phenyl-1,2-dihydroquinoline-3-carboxamide 5-Chloro-4-hydroxy-1-Methyl-2-oxo-1,2-dihydro-quinoline-3-carboxylic acid ethyl-phenyl-aMide LAQUINIMOD,5-CHLORO-4-HYDROXY-1-METHYL-2-OXO-1,2-DIHYDRO-QUINOLINE-3-CARBOXYLIC ACID ETHYL-PHENYL-AMIDE |
| CAS | 248281-84-7 |
| EINECS | 692-155-1 |
| InChI | InChI=1/C19H17ClN2O3/c1-3-22(12-8-5-4-6-9-12)19(25)16-17(23)15-13(20)10-7-11-14(15)21(2)18(16)24/h4-11,23H,3H2,1-2H3 |
248281-84-7 - Physico-chemical Properties
| Molecular Formula | C19H17ClN2O3 |
| Molar Mass | 356.8 |
| Density | 1.395±0.06 g/cm3(Predicted) |
| Melting Point | 201 °C (decomp) |
| Boling Point | 484.8±45.0 °C(Predicted) |
| Flash Point | 246.998°C |
| Solubility | DMSO 61 mg/mL (170.96 mM);Water <1 mg/mL (<1 mM);Ethanol 1 mg/mL (2.8 mM) |
| Vapor Presure | 0mmHg at 25°C |
| Appearance | solid |
| Color | Off-white |
| pKa | 4.50±1.00(Predicted) |
| Storage Condition | Sealed in dry,Room Temperature |
| Stability | Stable for 2 years from date of purchase as supplied. Solutions in DMSO may be stored at -20°C for up to 3 months. |
| Refractive Index | 1.674 |
| Use | Laquinimod are effective immunomodulators. Phase 3. |
| In vitro study | 0.1-1 μm Laquinimod treatment did not affect peripheral blood mononuclear cell (PBMC) activity. Large-scale gene expression microarray analysis in PBMC of healthy subjects and relapsing-remitting multiple sclerosis (RRMS) patients showed that Laquinimod inhibits the expression of genes associated with antigen presentation and corresponding inflammatory pathways. Laquinimod acts on CD14 |
| In vivo study | Laquinimod treated Lewis rats at a dose of 0.16-16 mg/kg per day to inhibit the occurrence of experimental autoimmune neuritis (EAN), improve clinical signs, and inhibit P0 peptide 180-199 specific T cell responses, and the demyelination of peripheral nerve inflammation, indicating that Laquinimod can regulate the effect by regulating the balance of Th1/Th2 cytokines. Laquinimod significantly inhibited the development of acute experimental autoimmune encephalomyelitis (EAE) in mice, 20 times more effective than the immunomodulator Roquinimex. Laquinimod acts on Lewis rats to inhibit the development of experimental autoimmune encephalomyelitis (EAE) in a dose-dependent manner and is better than Roquinimex (Linomide). Laquinimod acts on IFN-beta K. O. Mice and wild-type mice and effectively inhibits the development of chronic experimental autoimmune encephalomyelitis (chEAE). Laquinimod acts on C57BL/6 mice carrying MOG(35-55) peptide-induced active EAE to reduce clinical signs, inflammation and demyelination, and down-regulate VLA-4-Regulated adhesion and pro-inflammatory cytokines, such as IL-17. The effect of Laquinimod on the EAE mouse model was studied using a conditional BDNF knockout strain lacking BDNF expression in bone marrow cells and T cells (LLF mice), this indicates that Laquinimod also regulates autoimmune demyelination by inducing brain-driven neurotrophic factor (BDNF). |
248281-84-7 - Risk and Safety
| Hazard Symbols | T - Toxic |
| Risk Codes | 25 - Toxic if swallowed |
| Safety Description | 45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) |
| UN IDs | UN 2811 6.1 / PGIII |
248281-84-7 - Preparation solution concentration reference
| 1mg | 5mg | 10mg | |
|---|---|---|---|
| 1 mM | 2.803 ml | 14.013 ml | 28.027 ml |
| 5 mM | 0.561 ml | 2.803 ml | 5.605 ml |
| 10 mM | 0.28 ml | 1.401 ml | 2.803 ml |
| 5 mM | 0.056 ml | 0.28 ml | 0.561 ml |
Last Update:2024-01-02 23:10:35
248281-84-7 - Reference Information
| Use | Laquinmod is an immunomodulatory drug jointly developed by Teva Pharmaceutical Company and Active Biotechnology. It is mainly used to treat multiple sclerosis. Compared with other drugs for multiple sclerosis such as interferon and mitoxantrone, it is convenient to use and has weaker adverse reactions. |
| Preparation | Use 2-amino-6-chlorobenzoic acid to react with phosgene to form a ring to generate 5-chloroetinic anhydride (1), and then react with methyl iodide under the action of sodium hydride to obtain N-methyl-5-chloroetinic anhydride (2), compound 2 is condensed with diethyl malonate to obtain 5-chloro-1, 2-Dihydro-4-hydroxy-1-methyl-2-oxo-3-quinoline ethyl formate (3), compound 3 is hydrolyzed under HCl conditions to obtain 5-chloro-1, 2-dihydro-4-hydroxy-1-methyl-2-oxo-3-quinolinic acid (4), compound 4 is amidated with N-ethylaniline to obtain the target compound 5, the synthetic route is shown in Figure 1. In the synthesis of compound 1, the literature uses phosgene as a ring-forming reaction reagent, but because phosgene is too toxic and not suitable for industrial production, it is changed to solid phosgene. At the same time, when synthesizing compound 4, consider its reaction mechanism, The reaction feeding sequence and the amount of reaction substances have an important influence on the reaction. When synthesizing the compound laquinimod, the effects of different acid binding agents on the reaction were investigated. In this experiment, pyridine was finally determined as an acid binding agent. The synthetic route of laquinimod |
| biological activity | Laquinimod (ABR-215062, LAQ) is an effective immunomodulator. Phase 3. |
Last Update:2024-04-09 19:05:11
Supplier List
Spot supply
Product Name: Laquinimod Visit Supplier Webpage Request for quotationCAS: 248281-84-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: CIVENTICHEM CV-4057 Request for quotation
CAS: 248281-84-7
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 248281-84-7
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Spot supply
Product Name: Laquinimod Visit Supplier Webpage Request for quotationCAS: 248281-84-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Laquinimod Visit Supplier Webpage Request for quotationCAS: 248281-84-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: CIVENTICHEM CV-4057 Request for quotation
CAS: 248281-84-7
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 248281-84-7
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Spot supply
Product Name: Laquinimod Visit Supplier Webpage Request for quotationCAS: 248281-84-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


