247564-72-3
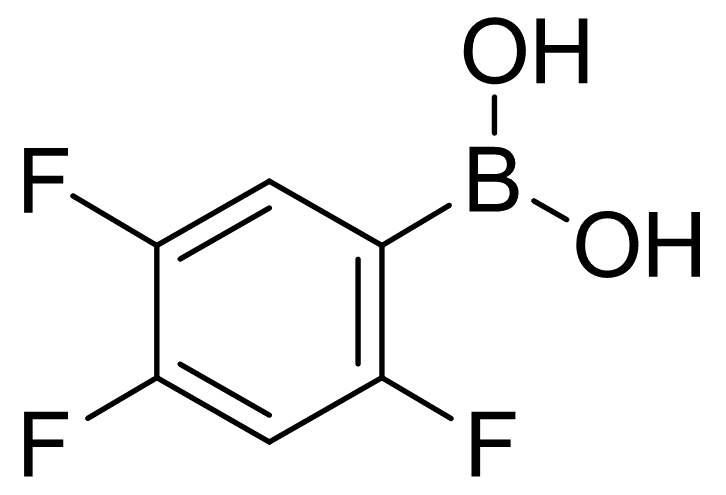
2,4,5-Trifluorobenzeneboronic acid
CAS: 247564-72-3
Molecular Formula: C6H4BF3O2
247564-72-3 - Names and Identifiers
| Name | 2,4,5-Trifluorobenzeneboronic acid |
| Synonyms | 2,4,5-Trifluorophen RARECHEM AH PB 0119 2,4,5-TRIFLUOROPHENYLBORONIC ACID 2,4,5-Trifluorophenylboronic acid 2,4,5-TRIFLUOROBENZENEBORONIC ACID 2,4,5-Trifluorobenzeneboronic acid 2,4,5-Trifluorophenylboronic Aicd (2,4,5-Trifluorophenyl)boronic acid Boronic acid, B-(2,4,5-trifluorophenyl)- |
| CAS | 247564-72-3 |
| InChI | InChI=1/C6H4BF3O2/c8-4-2-6(10)5(9)1-3(4)7(11)12/h1-2,11-12H |
247564-72-3 - Physico-chemical Properties
| Molecular Formula | C6H4BF3O2 |
| Molar Mass | 175.9 |
| Density | 1.44±0.1 g/cm3(Predicted) |
| Melting Point | 100°C(lit.) |
| Boling Point | 257.0±50.0 °C(Predicted) |
| Flash Point | 109.2°C |
| Vapor Presure | 0.00768mmHg at 25°C |
| Appearance | White to light yellow crystal powder |
| pKa | 7.38±0.58(Predicted) |
| Storage Condition | Room Temprature |
| Refractive Index | 1.465 |
| MDL | MFCD01863165 |
247564-72-3 - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| Risk Codes | 36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. S36 - Wear suitable protective clothing. S37 - Wear suitable gloves. |
| WGK Germany | 3 |
| HS Code | 29319090 |
| Hazard Class | IRRITANT |
247564-72-3 - Introduction
2,4,5-Trifluorobenzeneboronic acid is an organic compound with the chemical formula C6H3BF3O2. It is a solid, soluble in some organic solvents, such as dimethylformamide and dichloromethane.
2,4,5-Trifluorobenzeneboronic acid has many applications in chemical research and organic synthesis. It is an important Organoboron reagent, widely used in metal-catalyzed reactions, such as bonding reactions, cross-coupling reactions and Suzuki-Miyaura reactions. It can also be used as a fluorescent label, a complexing reagent, and an intermediate for organic optoelectronic materials.
The method for preparing 2,4,5-Trifluorobenzeneboronic acid is mainly obtained by reacting phenyl boronic acid with trifluorocarbonate. The reaction process generally needs to be carried out under an inert atmosphere and requires a certain catalyst. In addition, it can also be obtained by first preparing a complex of phenylboron trifluoride and 2,4, 5-trifluorocarbonic acid and carrying out a hydrolysis reaction under alkaline conditions.
Regarding safety information, 2,4,5-Trifluorobenzeneboronic acid is generally stable under normal conditions of use, but prolonged contact with skin and inhalation of dust or gas should be avoided. Wear appropriate protective gloves and goggles during handling. In case of ingestion or aspiration, seek medical attention immediately. In addition, during storage and handling, safe operating procedures should be observed and good ventilation conditions should be maintained.
2,4,5-Trifluorobenzeneboronic acid has many applications in chemical research and organic synthesis. It is an important Organoboron reagent, widely used in metal-catalyzed reactions, such as bonding reactions, cross-coupling reactions and Suzuki-Miyaura reactions. It can also be used as a fluorescent label, a complexing reagent, and an intermediate for organic optoelectronic materials.
The method for preparing 2,4,5-Trifluorobenzeneboronic acid is mainly obtained by reacting phenyl boronic acid with trifluorocarbonate. The reaction process generally needs to be carried out under an inert atmosphere and requires a certain catalyst. In addition, it can also be obtained by first preparing a complex of phenylboron trifluoride and 2,4, 5-trifluorocarbonic acid and carrying out a hydrolysis reaction under alkaline conditions.
Regarding safety information, 2,4,5-Trifluorobenzeneboronic acid is generally stable under normal conditions of use, but prolonged contact with skin and inhalation of dust or gas should be avoided. Wear appropriate protective gloves and goggles during handling. In case of ingestion or aspiration, seek medical attention immediately. In addition, during storage and handling, safe operating procedures should be observed and good ventilation conditions should be maintained.
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: 2,4,5-Trifluorophenylboronic Acid Visit Supplier Webpage Request for quotationCAS: 247564-72-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2,4,5-Trifluorophenylboronic acid Request for quotation
CAS: 247564-72-3
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 247564-72-3
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: 2,4,5-Trifluorophenylboronic Acid Visit Supplier Webpage Request for quotation
CAS: 247564-72-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 247564-72-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 2,4,5-Trifluorophenylboronic Acid Visit Supplier Webpage Request for quotationCAS: 247564-72-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2,4,5-Trifluorophenylboronic acid Request for quotation
CAS: 247564-72-3
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 247564-72-3
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: 2,4,5-Trifluorophenylboronic Acid Visit Supplier Webpage Request for quotation
CAS: 247564-72-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 247564-72-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


