247564-71-2
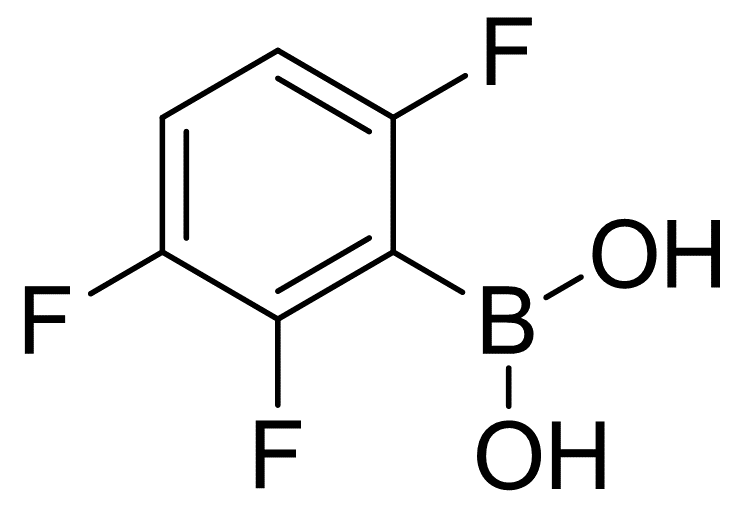
2,3,6-Trifluorophenylboronic acid
CAS: 247564-71-2
Molecular Formula: C6H4BF3O2
247564-71-2 - Names and Identifiers
| Name | 2,3,6-Trifluorophenylboronic acid |
| Synonyms | RARECHEM AH PB 0123 RARECHEM AH PB 0119 2,3,6-Trifluorophenylboronic acid 2,3,6-Trifluorobenzeneboronic acid (2,3,5-trifluorophenyl)boronic acid Boronic acid, B-(2,3,6-trifluorophenyl)- dimethyl(2-oxo-2-phenylethyl)sulfonium tetrafluoroborate |
| CAS | 247564-71-2 |
| InChI | InChI=1/C6H4BF3O2/c8-3-1-4(7(11)12)6(10)5(9)2-3/h1-2,11-12H |
247564-71-2 - Physico-chemical Properties
| Molecular Formula | C6H4BF3O2 |
| Molar Mass | 175.9 |
| Density | 1.44±0.1 g/cm3(Predicted) |
| Melting Point | 127-132 °C (lit.) |
| Boling Point | 266.0±50.0 °C(Predicted) |
| Flash Point | 116.7°C |
| Vapor Presure | 0.0036mmHg at 25°C |
| pKa | 7.09±0.58(Predicted) |
| Storage Condition | under inert gas (nitrogen or Argon) at 2-8°C |
| Refractive Index | 1.465 |
247564-71-2 - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| Risk Codes | 36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. S36 - Wear suitable protective clothing. |
| WGK Germany | 3 |
| Hazard Class | IRRITANT |
247564-71-2 - Introduction
2,3,6-Trifluorophenylboronic acid(2,3,6-Trifluorophenylboronic acid) is an organic compound with the chemical formula C6H4BF3O2. Its properties are as follows: colorless crystals, soluble in some organic solvents such as dimethyl sulfoxide and acetonitrile, insoluble in water.
2,3,6-Trifluorophenylboronic acid has a wide range of applications in the field of organic synthesis. It is an important aryl boronic acid derivative that can be used as a ligand to participate in organometallic-catalyzed reactions, such as fluorination catalyzed by Escherichia coli dehydrogenase. In addition, it can also be used in the synthesis of pharmaceutical and pesticide intermediates, or as a stable intermediate in organic synthesis.
There are two main methods for preparing 2,3,6-Trifluorophenylboronic acid. A common method is to react Phenylboronic acid with trifluorotoluene, and the resulting product is crystallized to give 2,3,6-Trifluorophenylboronic acid. Another method is to react 2,3, 6-trifluorophenyl alcohol with trichloroboryl acetate, followed by acid hydrolysis to obtain the target product.
Regarding the safety information of 2,3,6-Trifluorophenylboronic acid, since it is a chemical substance, appropriate safety measures need to be taken. It should be used in a well-ventilated laboratory environment and avoid contact with skin and eyes. Wear appropriate personal protective equipment such as lab gloves and goggles during operation. In addition, care should be taken to avoid inhaling its dust or gas. If ingested or inhaled, seek medical help immediately. When handling this compound, the relevant safety practices and regulations should be followed.
2,3,6-Trifluorophenylboronic acid has a wide range of applications in the field of organic synthesis. It is an important aryl boronic acid derivative that can be used as a ligand to participate in organometallic-catalyzed reactions, such as fluorination catalyzed by Escherichia coli dehydrogenase. In addition, it can also be used in the synthesis of pharmaceutical and pesticide intermediates, or as a stable intermediate in organic synthesis.
There are two main methods for preparing 2,3,6-Trifluorophenylboronic acid. A common method is to react Phenylboronic acid with trifluorotoluene, and the resulting product is crystallized to give 2,3,6-Trifluorophenylboronic acid. Another method is to react 2,3, 6-trifluorophenyl alcohol with trichloroboryl acetate, followed by acid hydrolysis to obtain the target product.
Regarding the safety information of 2,3,6-Trifluorophenylboronic acid, since it is a chemical substance, appropriate safety measures need to be taken. It should be used in a well-ventilated laboratory environment and avoid contact with skin and eyes. Wear appropriate personal protective equipment such as lab gloves and goggles during operation. In addition, care should be taken to avoid inhaling its dust or gas. If ingested or inhaled, seek medical help immediately. When handling this compound, the relevant safety practices and regulations should be followed.
Last Update:2024-04-09 21:54:55
Supplier List
Spot supply
Product Name: Atogepant Impurity 10 Visit Supplier Webpage Request for quotationCAS: 247564-71-2
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Spot supply
Product Name: 2,3,6-Trifluorophenylboronic acid Visit Supplier Webpage Request for quotationCAS: 247564-71-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2,3,6-Trifluorophenylboronic acid Request for quotation
CAS: 247564-71-2
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 247564-71-2
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: 2,3,6-Trifluorophenylboronic Acid Visit Supplier Webpage Request for quotation
CAS: 247564-71-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 247564-71-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Atogepant Impurity 10 Visit Supplier Webpage Request for quotationCAS: 247564-71-2
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Spot supply
Product Name: 2,3,6-Trifluorophenylboronic acid Visit Supplier Webpage Request for quotationCAS: 247564-71-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2,3,6-Trifluorophenylboronic acid Request for quotation
CAS: 247564-71-2
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 247564-71-2
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: 2,3,6-Trifluorophenylboronic Acid Visit Supplier Webpage Request for quotation
CAS: 247564-71-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 247564-71-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



