2427-71-6
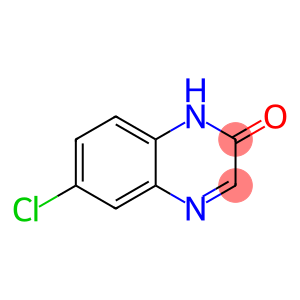
2-Hydroxy-6-Chloroquinoxaline
CAS: 2427-71-6
Molecular Formula: C8H5ClN2O
2427-71-6 - Names and Identifiers
| Name | 2-Hydroxy-6-Chloroquinoxaline |
| Synonyms | 6-Chloroquinoxalin-2-ol 6-chloro-2(1h)-quinoxalinon 6-CHLORO-2(1H)-QUINOXALINONE 6-chloroquinoxalin-2(1H)-one 6-Chloroquinoxaline-2(1H)-one 6-CHLORO-2-HYDROXYQUINOXALINE 2-Hydroxy-6-Chloroquinoxaline 2-HYDROXY-6-CHLOROQUINOXALINE 2(1H)-Quinoxalinone,6-chloro- 2(1H)-Quinoxalinone, 6-chloro- |
| CAS | 2427-71-6 |
| InChI | InChI=1/C8H5ClN2O/c9-5-1-2-6-7(3-5)10-4-8(12)11-6/h1-4H,(H,11,12) |
2427-71-6 - Physico-chemical Properties
| Molecular Formula | C8H5ClN2O |
| Molar Mass | 180.59 |
| Density | 1.50±0.1 g/cm3(Predicted) |
| Melting Point | 305 °C |
| Boling Point | 160-170 °C(Press: 10 Torr) |
| Flash Point | 193.6°C |
| Vapor Presure | 7.46E-07mmHg at 25°C |
| Appearance | Crystalline powder |
| pKa | 8.10±0.70(Predicted) |
| Storage Condition | Sealed in dry,Room Temperature |
| Refractive Index | 1.688 |
| Physical and Chemical Properties | This product is a white crystal, industrial light brown solid powder, difficult to dissolve in water, and alkali salt formation. |
2427-71-6 - Reference Information
| EPA chemical information | Information provided by: ofmpub.epa.gov (external link) |
| uses | 2-quinoxalol compounds such as 2-hydroxy-6-chloroquinoxalines are well-known intermediates in the production of medicinal chemicals and agrochemicals. 2-Hydroxy-6-chloroquinoxaline can be prepared from p-chloro-nitroacetanilide to quinoxalin-4-oxide and then reduced. 2-Hydroxy-6-chloroquinoxaline is an intermediate of the herbicide quinolone. |
| production method | its preparation method is to add N-4-chloro -2-nitrophenyl acetylacetamide and solvent to the reaction kettle, heat under reduced pressure and reflux for 1.5h, cool to room temperature, transfer to the reduction kettle, add the reducing agent, the reducing agent can be sodium borohydride or sodium bisulfite, and reduce at room temperature for 1.5h, transfer to a neutralization kettle for neutralization to pH = 4, then filter, wash the filter cake to neutral, and dry to obtain the finished product. |
Last Update:2024-04-09 02:00:07
Supplier List
Spot supply
Product Name: 2-Hydroxy-6-Chloroquinoxaline Visit Supplier Webpage Request for quotationCAS: 2427-71-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-Hydroxy-6-chloroquinoxaline Request for quotation
CAS: 2427-71-6
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 2427-71-6
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: 2-Hydroxy-6-Chloroquinoxaline Visit Supplier Webpage Request for quotation
CAS: 2427-71-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 2427-71-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 2-Hydroxy-6-Chloroquinoxaline Visit Supplier Webpage Request for quotationCAS: 2427-71-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-Hydroxy-6-chloroquinoxaline Request for quotation
CAS: 2427-71-6
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 2427-71-6
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: 2-Hydroxy-6-Chloroquinoxaline Visit Supplier Webpage Request for quotation
CAS: 2427-71-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 2427-71-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


