2353-33-5
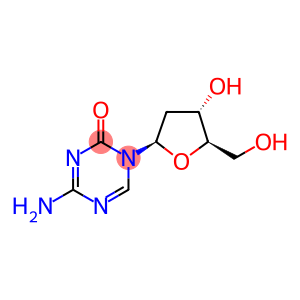
5-Aza-2'-deoxycytidine
CAS: 2353-33-5
Molecular Formula: C8H12N4O4
2353-33-5 - Names and Identifiers
| Name | 5-Aza-2'-deoxycytidine |
| Synonyms | Decitabine 5-azadeoxycytidine Decitabine(NSC127716) 5-Aza-2'-deoxycytidine 2'-desoxy-5-azacytidine Decitabine intermediates 4-amino-1-(2-deoxypentofuranosyl)-1,3,5-triazin-2(1H)-one 4-Amino-1-(2-deoxy-β-D-ribofuranosyl)-1,3,5-triazin-2(1H)-one 4-Amino-1-(2-deoxy-beta-D-erythro-pentofuranosyl)-1,3,5-triazin 4-amino-1-(2-deoxy-beta-d-erythro-pentofuranosyl)-s-triazin-2(1h)-on 4-amino-1-(2-deoxy-beta-d-erythro-pentofuranosyl)-s-triazin-2(1h)-one 4-Amino-1-(2-deoxy-β-D-erythro-pentofuranosyl)-1,3,5-triazin-2(1H)-one 5-triazin-2(1h)-one,4-amino-1-(2-deoxy-beta-d-erythro-pentofuranosyl)-3 4-amino-1-(2-deoxy-beta-D-erythro-pentofuranosyl)-1,3,5-triazin-2(1H)-one |
| CAS | 2353-33-5 |
| EINECS | 219-089-4 |
| InChI | InChI=1/C8H12N4O4/c9-7-10-3-12(8(15)11-7)6-1-4(14)5(2-13)16-6/h3-6,13-14H,1-2H2,(H2,9,11,15)/t4-,5+,6+/m0/s1 |
| InChIKey | XAUDJQYHKZQPEU-KVQBGUIXSA-N |
2353-33-5 - Physico-chemical Properties
| Molecular Formula | C8H12N4O4 |
| Molar Mass | 228.21 |
| Density | 1.3771 (rough estimate) |
| Melting Point | ~200°C (dec.) |
| Boling Point | 370.01°C (rough estimate) |
| Specific Rotation(α) | D22 +68.5° (30 min) +57.8° (6 hr) (c = 0.5 in water) |
| Flash Point | 247.6°C |
| Solubility | acetic acid/water (1:1): 50mg/mL |
| Vapor Presure | 1.78E-11mmHg at 25°C |
| Appearance | White crystalline powder |
| Color | White to Off-white |
| Merck | 14,2853 |
| BRN | 617982 |
| pKa | 14.02±0.60(Predicted) |
| Storage Condition | Keep in dark place,Inert atmosphere,Store in freezer, under -20°C |
| Stability | Stable. May be light or air sensitive. Incompatible with strong oxidizing agents. |
| Sensitive | Easily absorbing moisture |
| Refractive Index | 1.6590 (estimate) |
| MDL | MFCD00043011 |
| In vitro study | Decitabine effectively inhibited DNA synthesis in a dose-dependent manner, with IC50 values of 100 ng/mL and 1 ng/mL for HL-60 and KG1a leukemia cells, respectively. Decitabine inhibited cell growth in a dose-and time-dependent manner, with IC50 of about 100 ng/mL and 10 ng/mL for 72 and 96 hours of treatment of HL-60 and KG1a leukemia cells, respectively. The latest research shows that Decitabine acts on anaplastic large cell lymphoma (ALCL), has anti-proliferative and pro-apoptotic activities, and inhibits KARPAS-299 cells [ |
| In vivo study | Decitabine at 2.5 mg/kg dose for ALK |
2353-33-5 - Risk and Safety
| Risk Codes | R22 - Harmful if swallowed R36/37/38 - Irritating to eyes, respiratory system and skin. R68 - Possible risk of irreversible effects R61 - May cause harm to the unborn child |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36 - Wear suitable protective clothing. S36/37 - Wear suitable protective clothing and gloves. |
| WGK Germany | 3 |
| RTECS | XZ3012000 |
| FLUKA BRAND F CODES | 10-34 |
| HS Code | 29349990 |
| Toxicity | LD50 in mice (mg/kg): 190 i.p. (Momparler, 1985) |
2353-33-5 - Reference
| Reference Show more | 1. Hu Baocui Hao Jinqi Hou Ruili et al. Effects of 5-aza-2-deoxycytidine on CYP2E1 Gene Methylation and Protein Expression in ADIH Model Rats [J]. Journal of Baotou Medical College, 20202036 Volume 1, 36-39 Page 42 2020: national natural science foundation. 2. [IF = 4.24] Ling Guo et al."Purple sweet anthocyanins elicit calcium overload-induced cell death by inhibiting the calcium-binding protein S100A4 in acute lymphoblastic leukemia." Food Biosci. 2021 Aug;42:101214 |
Supplier List
Spot supply
Product Name: Decitabine Visit Supplier Webpage Request for quotationCAS: 2353-33-5
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Spot supply
Product Name: Decitabine Visit Supplier Webpage Request for quotationCAS: 2353-33-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: Decitabine Visit Supplier Webpage Request for quotationCAS: 2353-33-5
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Product Name: 5-Aza-2'-deoxycytidine Request for quotation
CAS: 2353-33-5
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 2353-33-5
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: 5-Aza-2'-deoxycytidine Request for quotation
CAS: 2353-33-5
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 2353-33-5
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
Spot supply
Product Name: 5-Aza-2′-deoxycytidine Visit Supplier Webpage Request for quotationCAS: 2353-33-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: 2'-DEOXY-5-AZACYTIDINE Request for quotation
CAS: 2353-33-5
Tel: 0512-67909115
Email:
Mobile: 18662510535
QQ: 99938187 Send QQ message
Send QQ message
CAS: 2353-33-5
Tel: 0512-67909115
Email:
Mobile: 18662510535
QQ: 99938187
Spot supply
Product Name: Decitabine Visit Supplier Webpage Request for quotationCAS: 2353-33-5
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Spot supply
Product Name: Decitabine Visit Supplier Webpage Request for quotationCAS: 2353-33-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: Decitabine Visit Supplier Webpage Request for quotationCAS: 2353-33-5
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Product Name: 5-Aza-2'-deoxycytidine Request for quotation
CAS: 2353-33-5
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 2353-33-5
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: 5-Aza-2'-deoxycytidine Request for quotation
CAS: 2353-33-5
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 2353-33-5
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
Spot supply
Product Name: 5-Aza-2′-deoxycytidine Visit Supplier Webpage Request for quotationCAS: 2353-33-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: 2'-DEOXY-5-AZACYTIDINE Request for quotation
CAS: 2353-33-5
Tel: 0512-67909115
Email:
Mobile: 18662510535
QQ: 99938187 Send QQ message
Send QQ message
CAS: 2353-33-5
Tel: 0512-67909115
Email:
Mobile: 18662510535
QQ: 99938187
View History





