2294-76-0
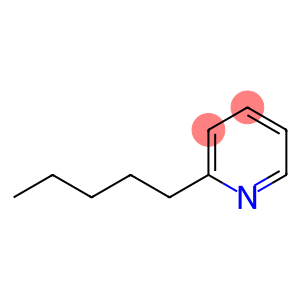
2-Pentylpyridine
CAS: 2294-76-0
Molecular Formula: C10H15N
2294-76-0 - Names and Identifiers
| Name | 2-Pentylpyridine |
| Synonyms | FEMA 3383 2-amylpyridine 2-Pentylpyridine FEMA NUMBER 3383 2-n-Amylpyridine 2-pentyl-pyridin 2-PENTYLPYRIDINE 2-N-AMYLPYRIDINE 2-Pentyl pyridine 2-N-PENTYLPYRIDINE 2-n-Pentylpyridine 1-(2-Pyridyl)pentane 1-(2-PYRIDYL)PENTANE |
| CAS | 2294-76-0 |
| EINECS | 218-937-0 |
| InChI | InChI=1/C10H15N/c1-2-3-4-7-10-8-5-6-9-11-10/h5-6,8-9H,2-4,7H2,1H3 |
2294-76-0 - Physico-chemical Properties
| Molecular Formula | C10H15N |
| Molar Mass | 149.23 |
| Density | 0.897 g/mL at 25 °C (lit.) |
| Melting Point | FDA 21 CFR (110) |
| Boling Point | 102-107 °C (lit.) |
| Flash Point | 175°F |
| JECFA Number | 1313 |
| Vapor Presure | 0.279mmHg at 25°C |
| Appearance | clear liquid |
| Specific Gravity | 0.902 |
| Color | Colorless to Light yellow |
| BRN | 2772 |
| pKa | 6.01±0.10(Predicted) |
| Storage Condition | Keep in dark place,Inert atmosphere,Room temperature |
| Refractive Index | n20/D 1.488(lit.) |
| MDL | MFCD00051828 |
| Physical and Chemical Properties | Colorless liquid, with veal-like aroma. Boiling point 102~107 deg C. Relative density (d420)0.881, refractive index (D20)1.4834. Slightly soluble in water, soluble in most commonly used organic solvents. Natural products are found in the low-boiling fraction of the steam distillation of light-fried beef and fried peanuts. |
2294-76-0 - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| Risk Codes | 36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36 - Wear suitable protective clothing. |
| WGK Germany | 3 |
| TSCA | Yes |
| HS Code | 29333990 |
| Hazard Note | Irritant |
2294-76-0 - Reference Information
| FEMA | 3383 | 2-PENTYLPYRIDINE |
| NIST chemical information | Information provided by: webbook.nist.gov (external link) |
| EPA chemical information | Information provided by: ofmpub.epa.gov (external link) |
| toxicity | GRAS(FEMA). |
| usage limit | FEMA(mg/kg): cold drinks, baked goods, jelly, pudding, 0.1; Seasonings, marinades, 0.4; Candy, meat products, meat soup, 0.5. |
| use | GB 2760-1996 specified as allowed food spices. |
| Production method | It is formed by alkylation of 2-pyridine methyl lithium. It is formed by hydrogenation of 2-pentenylpyridine. |
Last Update:2024-04-10 22:41:30
Supplier List
Spot supply
Product Name: 2-Amylpyridine Visit Supplier Webpage Request for quotationCAS: 2294-76-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-Pentylpyridine Request for quotation
CAS: 2294-76-0
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 2294-76-0
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: 2-Pentylpyridine Visit Supplier Webpage Request for quotation
CAS: 2294-76-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 2294-76-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 2-Amylpyridine Visit Supplier Webpage Request for quotationCAS: 2294-76-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-Pentylpyridine Request for quotation
CAS: 2294-76-0
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 2294-76-0
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: 2-Pentylpyridine Visit Supplier Webpage Request for quotation
CAS: 2294-76-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 2294-76-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


