201024-57-9
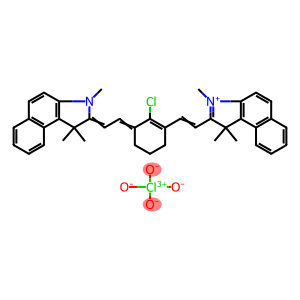
IR-813 perchlorate
CAS: 201024-57-9
Molecular Formula: C40H40Cl2N2O4
201024-57-9 - Names and Identifiers
201024-57-9 - Physico-chemical Properties
| Molecular Formula | C40H40Cl2N2O4 |
| Molar Mass | 683.66 |
| Melting Point | 238-242°C(lit.) |
| Storage Condition | Room Temprature |
201024-57-9 - Risk and Safety
| Safety Description | S22 - Do not breathe dust. S24/25 - Avoid contact with skin and eyes. |
201024-57-9 - Introduction
IR-813 perchlorate is an inorganic compound with the chemical formula (NH4) 2ircl6. The following is a description of the properties, uses, preparation and safety information of the compound:
Nature:
- IR-813 perchlorate is a yellow crystalline solid that dissolves in water.
-It has the characteristics of perchlorate, which contains hexavalent iridium ions (Ir6).
-It is a strong oxidizing agent that reacts with many reducing agents to release chlorine.
-It has good transparency in the optical field and is often used to prepare optical materials and coatings.
Use:
- IR-813 perchlorate is commonly used in the preparation of dyes, catalysts and electronic materials.
-It can be used as a standard substance for quantitative analysis in chemical analysis.
-In the optical field, IR-813 perchlorate is used to prepare transparent conductive films, such as ITO (indium tin oxide) films.
Method:
- IR-813 perchlorate can be prepared by reacting an aqueous solution of hexachloroammonium Iridium (NH4)2[IrCl6] with perchloric acid (HClO4).
-During the preparation process, attention should be paid to the selection of operating conditions and reactants.
Safety Information:
- IR-813 perchlorate is a toxic substance and should be operated under safe facilities.
-During use, avoid direct contact with skin, eyes or respiratory system, and maintain good ventilation.
-If contact occurs, rinse immediately with plenty of water and consult a doctor.
-When storing, keep away from fire and flammable materials.
Nature:
- IR-813 perchlorate is a yellow crystalline solid that dissolves in water.
-It has the characteristics of perchlorate, which contains hexavalent iridium ions (Ir6).
-It is a strong oxidizing agent that reacts with many reducing agents to release chlorine.
-It has good transparency in the optical field and is often used to prepare optical materials and coatings.
Use:
- IR-813 perchlorate is commonly used in the preparation of dyes, catalysts and electronic materials.
-It can be used as a standard substance for quantitative analysis in chemical analysis.
-In the optical field, IR-813 perchlorate is used to prepare transparent conductive films, such as ITO (indium tin oxide) films.
Method:
- IR-813 perchlorate can be prepared by reacting an aqueous solution of hexachloroammonium Iridium (NH4)2[IrCl6] with perchloric acid (HClO4).
-During the preparation process, attention should be paid to the selection of operating conditions and reactants.
Safety Information:
- IR-813 perchlorate is a toxic substance and should be operated under safe facilities.
-During use, avoid direct contact with skin, eyes or respiratory system, and maintain good ventilation.
-If contact occurs, rinse immediately with plenty of water and consult a doctor.
-When storing, keep away from fire and flammable materials.
Last Update:2024-04-10 22:29:15
Supplier List
Product Name: IR-813 PERCHLORATE Request for quotation
CAS: 201024-57-9
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 201024-57-9
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: IR-813 PERCHLORATE Request for quotation
CAS: 201024-57-9
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 201024-57-9
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
View History
