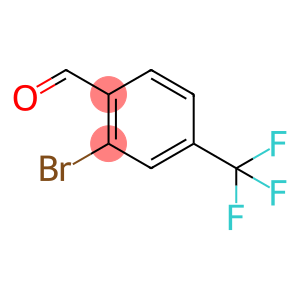
2-bromo-4-(trifluoromethyl)benzaldehyde
2-bromo-4-(trifluoromethyl)benzaldehyde
CAS: 85118-24-7
Molecular Formula: C8H4BrF3O
2-bromo-4-(trifluoromethyl)benzaldehyde - Names and Identifiers
| Name | 2-bromo-4-(trifluoromethyl)benzaldehyde |
| Synonyms | Einecs 285-677-2 3-Bromo-4-formylbenzotrifluoride -bromo-4-(trifluoromethyl)benzaldehyde 2-bromo-4-(trifluoromethyl)benzaldehyde Benzaldehyde, 2-bromo-4-(trifluoromethyl)- |
| CAS | 85118-24-7 |
| EINECS | 285-677-2 |
| InChI | InChI=1/C8H4BrF3O/c9-7-3-6(8(10,11)12)2-1-5(7)4-13/h1-4H |
2-bromo-4-(trifluoromethyl)benzaldehyde - Physico-chemical Properties
| Molecular Formula | C8H4BrF3O |
| Molar Mass | 253.02 |
| Density | 1.677±0.06 g/cm3 (20 ºC 760 Torr) |
| Boling Point | 230.0±40.0℃ (760 Torr) |
| Flash Point | 92.9±27.3℃ |
| Vapor Presure | 0.0676mmHg at 25°C |
| Storage Condition | under inert gas (nitrogen or Argon) at 2-8°C |
| Refractive Index | 1.517 |
2-bromo-4-(trifluoromethyl)benzaldehyde - Reference Information
| Use | 2-bromo-4-trifluoromethylbenzaldehyde is useful as an intermediate in organic synthesis. 2-bromo-4-trifluoromethylbenzaldehyde 2-bromo-4-(trifluoromethyl) benzoic acid can be prepared by oxidation of 2-bromo-1-methyl-4-trifluoromethylbenzene, it is then reduced to 2-bromo-4-(trifluoromethyl) phenyl] methane-1-ol, which is finally obtained by further oxidation. |
| preparation | 2-bromo-4-trifluoromethylbenzaldehyde was prepared as follows:(a) 2-bromo-4-(trifluoromethyl) benzoic acid: to 2-bromo-1-methyl-4-trifluoromethylbenzene (7.6g,32mmol,ABCR) in pyridine (75ml) to this solution, tetraethylammonium permanganate (24g,96mmol) was added and the reaction mixture was heated to 70 °c and stirred at this temperature for 30 hours. The reaction mixture was cooled to 25 °c and poured into an ice bath containing concentrated HCl(150ml) and NaHSO3(150g). The mixture became a clear aqueous solution and was extracted with EtOAc(4 x 200ml) and the combined extracts were washed with saturated NaCl(200ml), dried over Na2SO4, filtered and concentrated in vacuo to give the title product, white solid. MS(ESI, negative ion) m/z:267(M-1). (B) 2-bromo-4-(trifluoromethyl) phenyl] methan-1-ol: protected under N2, to a round-bottom flask equipped with a magnetic stirring and reflux condenser was added a solution of 2-bromo-4-(trifluoromethyl) benzoic acid (5.0g,24mmol,ABCR) in tetrahydrofuran at 0°C, 1.0m Borane-THF complex (72ML, 72mmol) was then added. The reaction mixture was heated to 65 °c and stirred at this temperature for 2 hours, the reaction mixture was cooled to 25 °c, and the solvent was removed in vacuo. The resulting residue was dissolved in CH2Cl2(100ml) and washed with saturated Na2CO3(100ml), the aqueous phase was back-extracted with CH2Cl2(4 x 80ml), the combined organic extracts were washed with saturated NaCl (200ml), dried over Na2SO4, filtered and concentrated in vacuo by Silica Gel chromatography (gradient: 0-4%EtOAc in hexanes) to give the title product as a colorless oil. (c) 2-bromo-4-trifluoromethylbenzaldehyde: to 2-bromo-4-(trifluoromethyl) phenyl] methan-1-ol (4.4g,23mmol) to a solution in CH2Cl2 (38.4 ml) was added pyridinium dichromate (g,102mmol,Fluka). The reaction mixture was stirred at 25 °c overnight and then filtered through Celite. The Celite pad was washed with CH2Cl2(2 x 50ml) and the combined filtrates were concentrated in vacuo. Purification by Silica Gel chromatography (gradient: 0-5%EtOAc in hexanes) afforded the title product 2-bromo-4-trifluoromethylbenzaldehyde (4.6g,18mmol) as a colorless oil. |
Last Update:2024-04-09 20:52:54
Supplier List
Spot supply
Product Name: 2-Bromo-4-(trifluoromethyl)benzaldehyde Visit Supplier Webpage Request for quotationCAS: 85118-24-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-bromo-4-(trifluoromethyl)benzaldehyde Request for quotation
CAS: 85118-24-7
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 85118-24-7
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: 2-bromo-4-(trifluoromethyl)benzaldehyde Visit Supplier Webpage Request for quotation
CAS: 85118-24-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 85118-24-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 2-Bromo-4-(trifluoromethyl)benzaldehyde Visit Supplier Webpage Request for quotationCAS: 85118-24-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-bromo-4-(trifluoromethyl)benzaldehyde Request for quotation
CAS: 85118-24-7
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 85118-24-7
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: 2-bromo-4-(trifluoromethyl)benzaldehyde Visit Supplier Webpage Request for quotation
CAS: 85118-24-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 85118-24-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


