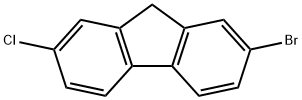
2-bromine-7-chlorinefluorene
2-bromine-7-chlorinefluorene
CAS: 99586-26-2
Molecular Formula: C13H8BrCl
2-bromine-7-chlorinefluorene - Names and Identifiers
| Name | 2-bromine-7-chlorinefluorene |
| Synonyms | 2-Bromo-7-Chlorofluorene 2-bromine-7-chlorinefluorene 2-bromine-7-chlorine fluorene 9H-Fluorene, 2-bromo-7-chloro- |
| CAS | 99586-26-2 |
2-bromine-7-chlorinefluorene - Physico-chemical Properties
| Molecular Formula | C13H8BrCl |
| Molar Mass | 279.56 |
| Density | 1.584±0.06 g/cm3(Predicted) |
| Melting Point | 156-157 °C |
| Boling Point | 386.8±35.0 °C(Predicted) |
2-bromine-7-chlorinefluorene - Introduction
It is an organic compound containing bromine and chlorine atoms. It has a chemical formula of C13H8BrCl and a molecular weight of 277.56g/mol.
Regarding the nature, it is a solid substance with a white crystalline appearance. It is almost insoluble in water at room temperature, but can be dissolved in many organic solvents such as ethanol, acetone and chloroform. It has a relatively high melting and boiling point, so it may decompose at high temperatures.
In terms of use, it is an important organic synthesis intermediate. It can be used to synthesize other compounds, such as organic photoelectric materials, organic semiconductors and drugs in the field of medicine. It can also be used as a reagent and catalyst.
The method of preparing fluorene is usually by reacting 2-bromo-7-fluorene and chlorine in an appropriate solvent. The specific preparation method can be adjusted according to specific requirements and conditions.
As for safety information, such as any chemicals, you need to follow proper laboratory and safety procedures when using the product. It can be irritating and injurious to the skin, eyes and respiratory system, so wear appropriate personal protective equipment such as gloves, goggles and protective masks. Avoid inhalation, ingestion or contact with skin and maintain good ventilation when handling this compound. Avoid fire and high temperatures during storage and handling. If an accident or discomfort occurs, you should immediately seek help from a medical professional.
Important note: When conducting any chemical experiments or using chemicals, always use personal protective equipment and follow safe operating procedures. For specific preparation methods and safety information, please refer to the relevant literature or consult a professional.
Regarding the nature, it is a solid substance with a white crystalline appearance. It is almost insoluble in water at room temperature, but can be dissolved in many organic solvents such as ethanol, acetone and chloroform. It has a relatively high melting and boiling point, so it may decompose at high temperatures.
In terms of use, it is an important organic synthesis intermediate. It can be used to synthesize other compounds, such as organic photoelectric materials, organic semiconductors and drugs in the field of medicine. It can also be used as a reagent and catalyst.
The method of preparing fluorene is usually by reacting 2-bromo-7-fluorene and chlorine in an appropriate solvent. The specific preparation method can be adjusted according to specific requirements and conditions.
As for safety information, such as any chemicals, you need to follow proper laboratory and safety procedures when using the product. It can be irritating and injurious to the skin, eyes and respiratory system, so wear appropriate personal protective equipment such as gloves, goggles and protective masks. Avoid inhalation, ingestion or contact with skin and maintain good ventilation when handling this compound. Avoid fire and high temperatures during storage and handling. If an accident or discomfort occurs, you should immediately seek help from a medical professional.
Important note: When conducting any chemical experiments or using chemicals, always use personal protective equipment and follow safe operating procedures. For specific preparation methods and safety information, please refer to the relevant literature or consult a professional.
Last Update:2024-04-10 22:30:50
Supplier List
Product Name: 2-Bromo-7-Chlorofluorene Request for quotation
CAS: 99586-26-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 99586-26-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Product Name: 2-Bromo-7-Chlorofluorene Request for quotation
CAS: 99586-26-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 99586-26-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
View History
