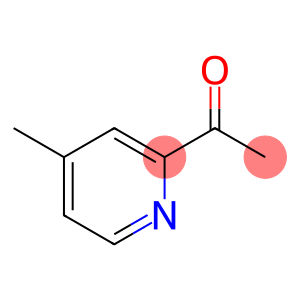
2-acetyl-4-methylpyridine
2-acetyl-4-methylpyridine
CAS: 59576-26-0
Molecular Formula: C8H9NO
2-acetyl-4-methylpyridine - Names and Identifiers
| Name | 2-acetyl-4-methylpyridine |
| Synonyms | 2-Acetyl-4-picoline 2-ACETYL-4-METHYLPYRIDIN 2-ACETYL-4-METHYLPYRIDINE 2-acetyl-4-methylpyridine 2-Acetyl-4-Methylpiridine 4-methyl-2-acetylpyridine 1-(4-methylpyridin-2-yl)ethanone 1-(4-METHYL-PYRIDIN-2-YL)-ETHANONE 1-(4-methylpyridin-2-yl)ethan-1-one |
| CAS | 59576-26-0 |
| InChI | InChI=1/C8H9NO/c1-6-3-4-9-8(5-6)7(2)10/h3-5H,1-2H3 |
2-acetyl-4-methylpyridine - Physico-chemical Properties
| Molecular Formula | C8H9NO |
| Molar Mass | 135.16 |
| Density | 1.036±0.06 g/cm3(Predicted) |
| Melting Point | 30-34°C(lit.) |
| Boling Point | 95-97 °C(Press: 15 Torr) |
| Flash Point | 203°F |
| Solubility | soluble in Methanol |
| Vapor Presure | 0.0788mmHg at 25°C |
| Appearance | powder to lump |
| Color | White to Gray to Brown |
| pKa | 3.82±0.10(Predicted) |
| Storage Condition | Keep in dark place,Sealed in dry,Room Temperature |
| Refractive Index | 1.512 |
| MDL | MFCD01863638 |
2-acetyl-4-methylpyridine - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| Risk Codes | 36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36 - Wear suitable protective clothing. |
| WGK Germany | 3 |
| HS Code | 29333990 |
2-acetyl-4-methylpyridine - Reference Information
| Application | 2-acetyl-4-methylpyridine is an organic intermediate with colorless needle-like crystals. 2-acetyl-4-methylpyridine can be obtained from the reaction of 2-nitrile-4-methylpyridine with methylmagnesium iodide or from the reaction of 4-methylpyridine with triacetaldehyde. |
| preparation | 4-methylpyridine (20g,215 mmol), trifluoroacetic acid (TFA,24.5g,215 mmol), tert-butyl hydroperoxide (53.4 mL of a 70 wt% aqueous solution, 415 mmol,Acros), iron (II) sulfate heptahydrate (1.0g,3.60mmol) A solution of triacetaldehyde (142g,1.07 mol) and MeCN(120 mL) was heated under reflux for 4 h. The reaction mixture was concentrated by distillation, cooled to room temperature, neutralized with saturated aqueous Na2CO3 solution, and extracted twice with EtOAc. The combined organic layers were washed with brine, dried over MgSO4, and concentrated in vacuo. The residue was purified by flash chromatography (EtOAc: n-hexane = 1: 4) to give the title compound as colorless needles in yield (5.80g,20%). |
Last Update:2024-04-09 21:01:54
Supplier List
Product Name: 2-Acetyl-4-methylpyridine Request for quotation
CAS: 59576-26-0
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 59576-26-0
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Spot supply
Product Name: 2-Acetyl-4-methylpyridine Visit Supplier Webpage Request for quotationCAS: 59576-26-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-Acetyl-4-methylpyridine Visit Supplier Webpage Request for quotation
CAS: 59576-26-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 59576-26-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: 2-Acetyl-4-methylpyridine Request for quotation
CAS: 59576-26-0
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 59576-26-0
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Spot supply
Product Name: 2-Acetyl-4-methylpyridine Visit Supplier Webpage Request for quotationCAS: 59576-26-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-Acetyl-4-methylpyridine Visit Supplier Webpage Request for quotation
CAS: 59576-26-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 59576-26-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


