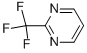
2-Trifluoromethylpyrimidine
2-Trifluoromethylpyrimidine
CAS: 116470-67-8
Molecular Formula: C5H3F3N2
2-Trifluoromethylpyrimidine - Names and Identifiers
| Name | 2-Trifluoromethylpyrimidine |
| Synonyms | 2-Trifluoromethylpyrimidine 2-Trifluoromethyl pyrimidine Pyrimidine, 2-(trifluoromethyl)- Pyrimidine, 2-(trifluoromethyl)- (9CI) |
| CAS | 116470-67-8 |
| EINECS | 946-092-4 |
2-Trifluoromethylpyrimidine - Physico-chemical Properties
| Molecular Formula | C5H3F3N2 |
| Molar Mass | 148.09 |
| Density | 1.353±0.06 g/cm3(Predicted) |
| Boling Point | 69.0±40.0 °C(Predicted) |
| pKa | -1.90±0.13(Predicted) |
| Storage Condition | Sealed in dry,Room Temperature |
2-Trifluoromethylpyrimidine - Reference Information
| use | 2-trifluoromethylpyrimidine is a very important organic intermediate, widely used in printing and dyeing, agriculture and other industries. |
| preparation | 1. preparation of 2-trichloromethylpyrimidine add 154mL 2-methylpyrimidine and 2.5g catalyst into a 250mL three-mouth bottle, mix evenly, heat in an oil bath, and start reflux. The initial reaction temperature is 100 ℃, the 500W iodine tungsten lamp is placed 35cm away from the reaction bottle, and chlorine gas is introduced from the bottom of the reactor under light. The chlorine gas speed is controlled by observing the color depth of the gas in the reaction tube, and it is appropriate to be light green under lamp irradiation. With the continuous increase of chlorination degree, the temperature of the reaction solution should be increased, but the reaction temperature should not exceed 150 ℃ until the end of the reaction. The experimental setup is shown in Figure 1. The reaction solution was alkaline washed with 10% sodium carbonate aqueous solution, then washed to neutral, anhydrous calcium chloride was added to dry to remove water, and the fraction was collected by decompression distillation at 97~98 ℃/1 .6kPa to obtain the product 2-trichloromethylpyrimidine for quantitative analysis. 2. Preparation of 2-trifluoromethylpyrimidine It is obtained by the interaction of 2-trichloromethylpyrimidine and anhydrous hydrogen fluoride. The molar ratio of 2-trichloromethylpyrimidine and anhydrous hydrogen fluoride is 1:3.88, and the reaction is fully reacted at a temperature of 104 ℃ and a pressure of 1.67-1.77MPa for 3 hours. The yield was 72.1%. Because anhydrous hydrogen fluoride is cheap and easy to obtain, the equipment is easy to solve, no special steel is required, the cost is low, and it is suitable for industrialization. |
Last Update:2024-04-09 20:52:54
Supplier List
Spot supply
Product Name: 2-(Trifluoromethyl)pyrimidine Visit Supplier Webpage Request for quotationCAS: 116470-67-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-Trifluoromethylpyrimidine Request for quotation
CAS: 116470-67-8
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 116470-67-8
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: 2-Trifluoromethylpyrimidine Visit Supplier Webpage Request for quotation
CAS: 116470-67-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 116470-67-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 2-(Trifluoromethyl)pyrimidine Visit Supplier Webpage Request for quotationCAS: 116470-67-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-Trifluoromethylpyrimidine Request for quotation
CAS: 116470-67-8
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 116470-67-8
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: 2-Trifluoromethylpyrimidine Visit Supplier Webpage Request for quotation
CAS: 116470-67-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 116470-67-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


