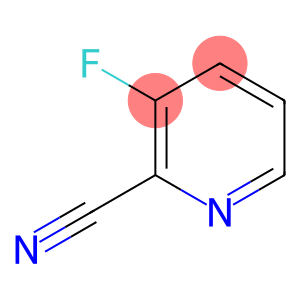
2-Cyano-3-fluoropyridine
2-Cyano-3-fluoropyridine
CAS: 97509-75-6
Molecular Formula: C6H3FN2
2-Cyano-3-fluoropyridine - Names and Identifiers
2-Cyano-3-fluoropyridine - Physico-chemical Properties
| Molecular Formula | C6H3FN2 |
| Molar Mass | 122.1 |
| Density | 1.24±0.1 g/cm3(Predicted) |
| Melting Point | 27-30°C |
| Boling Point | 120-125 °C(Press: 17 Torr) |
| Flash Point | 104°C |
| Solubility | Chloroform (Sparingly), DMSO (Slightly) |
| Appearance | Solid |
| Color | White to Off-White Low-Melting |
| pKa | -2.74±0.10(Predicted) |
| Storage Condition | Inert atmosphere,Room Temperature |
| Physical and Chemical Properties |
|
2-Cyano-3-fluoropyridine - Risk and Safety
| Risk Codes | R20/21/22 - Harmful by inhalation, in contact with skin and if swallowed. R36/37/38 - Irritating to eyes, respiratory system and skin. R41 - Risk of serious damage to eyes R37/38 - Irritating to respiratory system and skin. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. S23 - Do not breathe vapour. |
| UN IDs | 3276 |
| WGK Germany | 3 |
| HS Code | 29333990 |
| Hazard Note | Toxic |
| Hazard Class | 6.1 |
| Packing Group | III |
2-Cyano-3-fluoropyridine - Reference Information
| Application | 2-cyano-3-fluoropyridine is an organic intermediate, 2-cyano-3-fluoropyridine can be prepared from 3-fluoropyridine as a raw material in three steps or 3-fluoropyridine-N-oxide can be prepared by first oxidizing 3-fluoropyridine, it is then reacted with trimethylsilyl cyanide to give 2-cyano-3-fluoropyridine. |
| preparation | 3-fluoropyridin-n-oxide: 3-fluoropyridine (2.5G, 25.749mmol) dissolve in anhydrous DCM(75 ml). m-CPBA(70% suspension, 12.696G, 51.499mmol) was added and stirred overnight at room temperature. The reaction mixture was washed with saturated aqueous NaHCO3 and the organic phase was dried over Na2SO4, filtered and concentrated in vacuo. Purification by Silica Gel chromatography eluting with DCM and DCM/methanol (97: 3) gave the desired intermediate as a solid (1.413g,49%). 2-cyano-3-fluoropyridine: 3-fluoropyridine-N-oxide (1.0G, 8.687mmol) was dissolved in anhydrous acetonitrile (100 ml). Triethylamine (1.319G, 1.82 ML, 13.031mmol), trimethylsilyl cyanide (3.447G, 4.63 ML, 34.749mmol) were added and the mixture was heated to reflux, overnight. Cool to room temperature and concentrate in vacuo. The residue was dissolved in EtOAc and washed with saturated aqueous nahco3. The organic layer was dried over Na2SO4, filtered and concentrated in vacuo. Purification by Silica Gel chromatography eluting with hexane/EtOAc(1: 0 and 17: 3) gave the desired intermediate as a solid (746mg,70%). |
Last Update:2024-04-09 02:00:12
Supplier List
Spot supply
Product Name: 2-Cyano-3-fluoropyridine Visit Supplier Webpage Request for quotationCAS: 97509-75-6
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: 3-Fluoropicolinonitrile Visit Supplier Webpage Request for quotationCAS: 97509-75-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-Cyano-3-fluoropyridine Request for quotation
CAS: 97509-75-6
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 97509-75-6
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Spot supply
Product Name: 2-Cyano-3-fluoropyridine Visit Supplier Webpage Request for quotationCAS: 97509-75-6
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: 3-Fluoropicolinonitrile Visit Supplier Webpage Request for quotationCAS: 97509-75-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-Cyano-3-fluoropyridine Request for quotation
CAS: 97509-75-6
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 97509-75-6
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
View History


