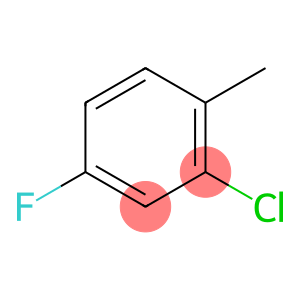
2-Chloro-4-fluorotoluene
2-Chloro-4-fluorotoluene
CAS: 452-73-3
Molecular Formula: C7H6ClF
2-Chloro-4-fluorotoluene - Names and Identifiers
| Name | 2-Chloro-4-fluorotoluene |
| Synonyms | oro-4-fL 2-Chlor-4-fluortoluol 4-FLUORO-2-CHLOROTOLUENE 2-CHLORO-4-FLUOROTOLUENE 2-Chloro-4-fluorotoluene 1-Brom-2-fluor-4-methylbenzol 1-bromo-2-fluoro-4-methylbenzene 2-CHLORO-4-FLUORO-1-METHYLBENZENE Benzene, 1-bromo-2-fluoro-4-methyl- Benzene, 2-chloro-4-fluoro-1-methyl- |
| CAS | 452-73-3 |
| EINECS | 207-209-8 |
| InChI | InChI=1/C7H6BrF/c1-5-2-3-6(8)7(9)4-5/h2-4H,1H3 |
| InChIKey | CSARJIQZOSVYHA-UHFFFAOYSA-N |
2-Chloro-4-fluorotoluene - Physico-chemical Properties
| Molecular Formula | C7H6ClF |
| Molar Mass | 144.57 |
| Density | 1.197 g/mL at 25 °C (lit.) |
| Boling Point | 154-156 °C (lit.) |
| Flash Point | 122°F |
| Vapor Presure | 0.942mmHg at 25°C |
| Appearance | clear liquid |
| Specific Gravity | 1.197 |
| Color | Colorless to Light yellow |
| BRN | 1931690 |
| Storage Condition | 2-8°C |
| Refractive Index | n20/D 1.499(lit.) |
| Physical and Chemical Properties | Density 1.19, boiling point 154-156 deg C, flash point 50 deg C. |
2-Chloro-4-fluorotoluene - Risk and Safety
| Risk Codes | R10 - Flammable R36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S16 - Keep away from sources of ignition. S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. S36 - Wear suitable protective clothing. |
| UN IDs | UN 1993 3/PG 3 |
| WGK Germany | 3 |
| HS Code | 29039990 |
| Hazard Note | Irritant/Flammable |
| Hazard Class | 3 |
| Packing Group | III |
2-Chloro-4-fluorotoluene - Reference Information
| NIST chemical information | Information provided by: webbook.nist.gov (external link) |
| application | 2-chloro-4-fluorotoluene can be synthesized into a variety of pesticide intermediates after photo-chlorination, nitrification, bromination, alkylation, reduction, etc. In recent years, with the rapid development of pesticide, medicine, dye and other industries, the demand for organic fluoride intermediates is increasing, and fluorobenzene chemicals have been widely used. |
| preparation | 1L reaction kettle (equipped with condenser, the diameter of the gas phase outlet is 1/6~1/5 of the diameter of the kettle) is cooled to below 20 ℃, 318g(15.9mol,15eq) of anhydrous hydrogen fluoride is added under stirring, and the temperature is lowered to below 5 ℃ after feeding, 150g(1.059mol,1eq) of 3-chloro -4-methylaniline was added dropwise, the temperature was controlled within the range of 5~15 ℃, the dropwise addition was completed for about 3h, and the heat preservation was continued for 2h. After heat preservation, the temperature is reduced to below 5 ℃, sodium nitrite (1.03eq) is added in batches, the reaction temperature is controlled in the range of -5~15 ℃, the material is added for about 4 hours, and the heat preservation is completed for 1 hour. After heat preservation, the temperature of the reactor is programmed (strictly control the temperature raising speed: in the range of 0~20 ℃, the temperature is 0.5~1 ℃ per hour, and in the range of 20~80 ℃, the temperature is 1~2 ℃ per hour), the temperature is raised to 80 ℃ and the temperature is kept for 2 hours. After heat preservation is completed, the temperature is reduced to 30-35 ℃, stratification is carried out, dilute alkali is added to the organic layer and neutralized to PH = 7-8, steam distillation is carried out, the fractions are stratified at room temperature to obtain 138g of organic layer, the purity detected by meteorological chromatography is 95.5%, 124g of qualified product is obtained by rectification, the yield is 81.0%, and the purity of 2-chlorine -4-fluorotoluene detected by meteorological chromatography is 99.9%. |
| use | pharmaceutical and pesticide intermediates. |
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: 2-Chloro-4-fluorotoluene Visit Supplier Webpage Request for quotationCAS: 452-73-3
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Product Name: 2-Chloro-4-fluorotoluene Request for quotation
CAS: 452-73-3
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 452-73-3
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
Spot supply
Product Name: 2-Chloro-4-fluorotoluene Visit Supplier Webpage Request for quotationCAS: 452-73-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-Chloro-4-fluorotoluene Request for quotation
CAS: 452-73-3
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 452-73-3
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
Spot supply
Product Name: 2-Chloro-4-fluorotoluene Visit Supplier Webpage Request for quotationCAS: 452-73-3
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Product Name: 2-Chloro-4-fluorotoluene Request for quotation
CAS: 452-73-3
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 452-73-3
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
Spot supply
Product Name: 2-Chloro-4-fluorotoluene Visit Supplier Webpage Request for quotationCAS: 452-73-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-Chloro-4-fluorotoluene Request for quotation
CAS: 452-73-3
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 452-73-3
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
View History



